Are Patients with Asthma and Chronic Obstructive Pulmonary Disease Preferred Targets of COVID-19?
Article information
Abstract
The coronavirus pandemic, known as coronavirus disease 2019 (COVID-19), is an infectious respiratory disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a novel coronavirus first identified in patients from Wuhan, China. Since December 2019, SARS-CoV-2 has spread swiftly around the world, infected more than 25 million people, and caused more than 800,000 deaths in 188 countries. Chronic respiratory diseases such as asthma and chronic obstructive pulmonary disease (COPD) appear to be risk factors for COVID-19, however, their prevalence remains controversial. In fact, studies in China reported lower rates of chronic respiratory conditions in patients with COVID-19 than in the general population, while the trend is reversed in the United States and Europe. Although the underlying molecular mechanisms of a possible interaction between COVID-19 and chronic respiratory diseases remain unknown, some observations can help to elucidate them. Indeed, physiological changes, immune response, or medications used against SARS-CoV-2 may have a greater impact on patients with chronic respiratory conditions already debilitated by chronic inflammation, dyspnea, and the use of immunosuppressant drugs like corticosteroids. In this review, we discuss importance and the impact of COVID-19 on asthma and COPD patients, the possible available treatments, and patient management during the pandemic.
Introduction
At the end of 2019, an outbreak of an atypical, viral and contagious pneumonia was reported in Wuhan, China, and a novel coronavirus was identified as the causative agent in January 2020 [1]. The novel coronavirus is closer to severe acute respiratory syndrome coronavirus (SARS-CoV)-1 than to Middle East respiratory syndrome coronavirus, which caused two major epidemics in 2002 and 2012, respectively. It has been provisionally named new coronavirus 2019 (nCoV-2019) [2], and then renamed SARS-CoV-2 by the International Virus Taxonomy Committee [3].
Known as coronavirus disease 2019 (COVID-19), the illness was characterized by varying signs and symptoms, ranging from a common infection of the upper respiratory tract to respiratory distress. Person-to-person transmission may occur, mainly via respiratory droplets; however, indirect transmission by droplets or aerosols of infected person deposited on inert surfaces remains uncertain [4]. COVID-19 quickly spread to reach the pandemic stage in March 2020 [5], affecting to date (August 31, 2020) more than 25 million people and causing 847,847 deaths worldwide, according to Johns Hopkins University.
SARS-CoV-2 seems to have preference for elderly populations, males or those with comorbidities. The scientific literature agrees that cardiovascular disease and diabetes are important risk factors for the contraction, morbidity and mortality of COVID-19, but diverge on the role of chronic respiratory diseases specifically between China, the United States and Europe. Chronic lung diseases are usually affected by respiratory infections of viral origin that may have an impact on their development and progression [6,7]. Indeed, chronic respiratory diseases, such as asthma and chronic obstructive pulmonary disease (COPD), have a multifactorial origin and are characterized by chronic inflammation of the airways that induces bronchial hyperresponsiveness and decreased lung function. In addition, infection with respiratory viruses including respiratory syncytial virus, rhinovirus or some coronaviruses (OC43, E229), induces significant physiological changes, mainly via the immune response, and may negatively impact patients’ respiratory tract and lead to exacerbation [8,9].
In this review, we analyze the risk of respiratory diseases during the SARS-CoV-2 pandemic and discuss the impact of COVID-19 on patients with chronic respiratory diseases including asthma and COPD.
Pathophysiology of SARS-CoV-2 Infection
COVID-19 is characterized by different symptoms that vary from one individual to another and from one study to another (Table 1) [10-16], including fever, dry cough, fatigue, myalgia, ageusia and anosmia [17], that can progress in the most serious cases to acute respiratory distress [18], specifically in patients with comorbidities [19]. The latter are considered as an important risk factor for the morbidity and mortality of COVID-19 [10]. Indeed, several diseases have been reported with varying frequency in patients infected with SARS-CoV-2 (Table 2) [10,19-29].
This single-stranded RNA virus belongs to the Coronaviridae family of the genus Betacoronavirus [30]. It has round or elliptic forms, with a diameter of approximately 60–140 nm [31] and shares the same life cycle as SARS-CoV-1. It binds specifically to the angiotensin converting enzyme (ACE)-2 and the transmembrane serine protease-2 of the host cell [32,33], via S glycoproteins “spike” present on its surface. The whole forms a complex, which allows the fusion of the two membranes [34] and the release of the viral genome in the cytoplasm [32] where it is translated into two polyproteins (pp1a and pp1ab) responsible for its replication and its transcription [31].
The newly synthesized genomic RNA, the core proteins, and the envelope glycoproteins assemble and form viral particles, contained in Golgi vesicles, which exit the cell by exocytosis to infect other cells and invade the mucous membranes of the airways [35].
Specialized immune cells recognize viral antigens and induce the activation of several signaling pathways and transcription factors [36] which, in turn, stimulate a variety of proinflammatory cytokines involved in the antiviral response by activating the phagocytic cells and mediating adaptive immunity [37,38]. T-helper (Th) lymphocytes, derived from the differentiation of activated CD4+ T lymphocytes, secrete cytokines that stimulate the differentiation of B lymphocytes into anti–SARS-CoV-2 antibody producing plasma cells, and CD8+ T lymphocytes to cytotoxic effector cells capable of eliminating infected cells [39,40], this results in lung lesions visible on computed tomography (CT)-scan [41]. In some patients the increased secretion of several cytokines causes pulmonary hyper-inflammation [20,42]; moreover, SARS-CoV-2 infection induces a decrease in the expression of ACE2, which, in turn, increases angiotensin 2 plasma concentrations that may cause worsening of respiratory symptoms or even acute respiratory distress [43].
COVID-19 and COPD
Evidence shows that a respiratory virus infection would impact patients with chronic respiratory diseases such as asthma and COPD; hence, lower respiratory infections are considered as important risk factors for exacerbation and hospitalization [44,45]. The World Health Organization (WHO) classifies asthma and COPD as major public health problems, in fact, COPD was the third leading cause of death worldwide in 2016 [46].
The fact is that studies do not show a correlation between having a chronic respiratory disease and contracting SARS-CoV-2 infection, but it represents one of the most reported comorbidities in COVID-19 patients [47]. Based on studies from China, Halpin et al. concluded that the prevalence of chronic respiratory disease in COVID-19 patients was significantly lower than its prevalence in the general population [48]. However, in the United States and Europe, it was at least as high, if not greater, than their prevalence in the general population [21,49,50]. In addition, the reported rates of COPD are significantly higher than those of asthma in China, while the trend is reversed in the United States (Table 3) [10,11,21-25,27,29,50-59]. These prevalence differences could be explained by an underdiagnosis and misreporting of respiratory disease in COVID-19 patients in China compared to the United States and other European countries [50,51], or due to patients’ behavior amid the pandemic in those countries. In addition, a similar trend in the prevalence of the two diseases was observed in the general population. Indeed, in China, the prevalence of COPD was estimated at 8.6% [60] while that of asthma was only 4.2% [61]; however, in the United States the prevalence of COPD was 5.9% [62], compared to 7.7% for asthma [49].
Although SARS-CoV-2 infects healthy people and those with chronic respiratory diseases in the same way, the latter present with more severe symptoms [10,63], higher probability of being admitted to intensive care [64], and a higher mortality rate compared to patients without comorbidities [22]. The mechanisms underlying these differences in the virulence of SARS-CoV-2 between patients with chronic respiratory diseases and patients without comorbidities are still debated by the scientific community, however, some observations give us clues to explore and to elucidate them (Figure 1).
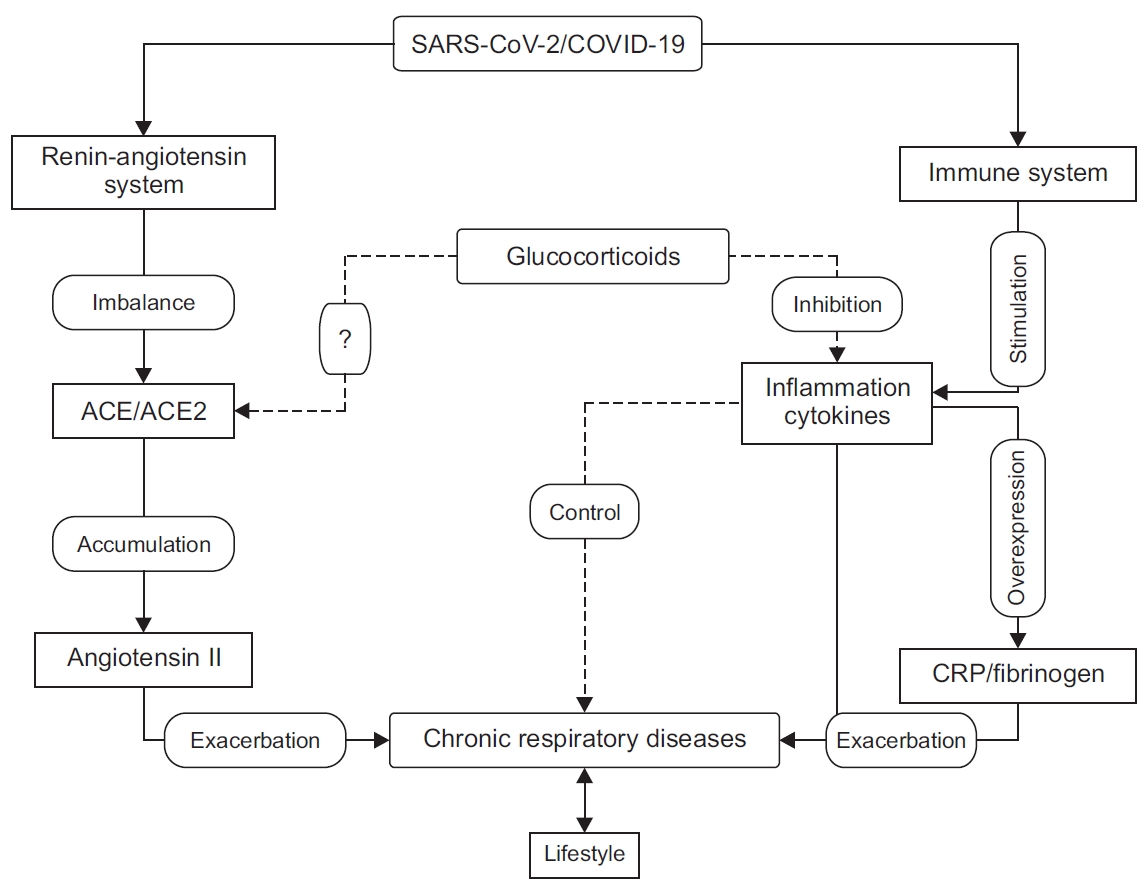
Impact of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) on chronic respiratory diseases. SARS-CoV-2 activates the host’s immune system and stimulates the production of pro-inflammatory cytokines, which may increase the risk of exacerbations of chronic respiratory diseases directly or indirectly by stimulating the production of C-reactive protein (CRP), fibrinogen, and cytokines. SARS-CoV-2 also affects the renin-angiotensin-aldosterone system by negatively regulating the expression of angiotensin-converting enzyme 2 (ACE2), which causes the accumulation of angiotensin 2 in the lung tissue and may exacerbate or worsen the symptoms of chronic respiratory diseases. Glucocorticoids (GCs) modulate the immune response and reduce inflammation and help to control chronic respiratory disease. The effect of GCs’ on ACE2 expression remains debated; their role in preventing SARS-CoV-2 infection has not been fully investigated. COVID-19: coronavirus disease 2019.
1. The renin-angiotensin aldosterone system
ACE2 is a transmembrane metalloprotease expressed in various tissues, including the heart, intestines, upper and lower airways [65]. This receptor is overexpressed in the epithelium of the bronchioles in COPD patients compared to controls, and could partly explain the severity of COVID-19 in these patients, since overexpression of ACE2 would facilitate virus penetration into the host cells [66]. By contrast, Jackson et al. reported that ACE2 is downregulated in atopic asthmatics after exposure to an allergen and suggested that this may explain the low prevalence of asthma in COVID-19 patients [67]. However, another study showed that there was no significant difference in the level of ACE2 expression throughout the bronchial epithelium in patients with chronic respiratory diseases and healthy controls [68]. Moreover, studies from the U.S. show a significantly higher prevalence of asthma sufferers with COVID-19 [23,24].
On the other hand, ACE is overexpressed in asthma and COPD patients [69]. Moreover, during SARS-CoV-2 infection, ACE2 is negatively regulated [70], leading to an increase in angiotensin 2 plasma concentrations, which are correlated with worsening of respiratory symptoms [71] and a high level of proinflammatory cytokines [72]. In brief, the ACE/ACE2 ratio seems to play an important role in modulating the immune response and the severity of COVID-19, particularly in patients with chronic respiratory diseases.
2. Biological parameters
Infection with SARS-CoV-2 leads to deregulation of some pro-inflammatory cytokine levels [11,42], and other biological parameters, some of which have been suggested to be important markers for predicting the severity of COVID-19. These include, among others, C-reactive protein (CRP), interleukin (IL)-6, lactate dehydrogenase, amyloid A protein, the ratio of neutrophils/lymphocytes, D-dimers and cardiac troponin, whose values are significantly higher in severe COVID-19 patients compared to non-severe patients. However, lymphocyte and platelet counts decrease with the severity of the disease [73]. This deregulation is independent of having a chronic respiratory disease [74], however, the consequences would not be the same for the patients with chronic respiratory disease compared to those without. In fact, the concentration of tumor necrosis factor alpha (TNF-α), which is higher in asthma patients compared to controls before infection [75], is positively correlated with bronchial hyperreactivity [76]. Moreover, a high TNF-α level is significantly related to a low maximal expiratory volume per second in asthma and COPD patients [77], thus perhaps increasing the risk of having an exacerbation during SARS-CoV-2 infection.
IL-6 is a pleiotropic cytokine involved in several inflammatory processes. It is also involved in modulating the immune response towards a Th-17 profile by initiating the differentiation of naive CD4 T cells into Th-17 pro-inflammatory effector cells, and by inhibiting their conversion into regulatory T cells that have the role of resolving the inflammation [78], thus maintaining the inflammatory state. Furthermore, a high level of IL-6 is linked to a deterioration of lung function [79] and an increase in mortality in COPD patients [80]. This could worsen the symptoms of COPD patients with COVID-19; indeed, COPD has been reported to be an independent risk factor for the severity of COVID-19 [63].
The increased serum level of IL-6 affects the liver and induces an overproduction of CRP and fibrinogen [81]. In COPD patients, these two proteins were found to be significantly higher than in controls [82]. The same trend was reported in asthmatics [83], where CRP level is inversely proportional to the asthma control test score [84]. In addition, increased CRP and fibrinogen concentrations were associated with more exacerbations and higher mortality rate in COPD patients [85]. In asthmatics, the exacerbation was accompanied by an increase of serum CRP level [86].
Although studies do not show a clear link between COVID-19 and exacerbation of chronic respiratory disease, other coronaviruses are risk factors [87], and the involved molecular mechanisms remain unknown. Nevertheless, some biological parameters induced by SARS-CoV-2 may constitute interesting targets to investigate.
3. Pneumonia
COVID-19 causes very pronounced pneumonia [88], in fact, the chest CT scans show unilateral or bilateral ground-glass opacities in almost all patients, including asymptomatic cases. These lung opacities progress to dense lesions at an advanced stage of the disease [10,41]; however, during the first 2 days after the onset of symptoms, the CT scan may be negative [89].
Pneumonia significantly impacts the length of hospital stay and increases the need for artificial respiration in COPD patients [90]. In asthmatics, pneumonia can increase the risk of being hospitalized or of being admitted to the emergency room for exacerbation during the following year after hospital discharge [91]. In addition, pneumoniae are linked to a decrease in the effectiveness of treatments used by asthmatics, specifically β-mimetics [92].
Prophylaxis, Treatment, and Management
1. Prevention
At present, prevention and protection measures are the only alternatives available to populations since no vaccine is expected for at least a year according to the WHO. A series of hygiene and preventive measures for the general population during the COVID-19 pandemic are published by WHO [93] and should be strictly applied, particularly by patients with chronic respiratory diseases as they are at high risk of developing severe symptoms when contracting SARS-CoV-2 infection.
2. Treatment and management
At this time, no specific treatment for COVID-19 is available; however, repositioning for some existing drugs is being tested around the world. Indeed, in China, the National Health Commission has advocated the use of existing antivirals: interferon-α commonly used to treat hepatitis-C, the lopinavir/ritonavir (Lpv/Rtv) combination used in the treatment of human immunodeficiency virus 1, Arbidol used to treat severe cases of influenza, chloroquine (CQ) and its derivatives (hydroxychloroquine [HCQ] and chloroquine-phosphate) known for their efficacy against plasmodium and Remdesivir that has a broad spectrum of antiviral action [94].
In South Korea, Lpv/Rtv was tested successfully on a 54-year-old patient with COVID-19, a significant drop in viral load and an improvement in the patients’ health were noticed [95]. However, a retrospective study suggested that this drug is more effective when combined with Arbidol [96]. This hypothesis was verified in another study where four patients were treated, three of whom were completely cured after 15 days of treatment, the fourth, who was in critical condition on admission, had his stay extended and his condition improved [97].
However, in a comparative study, Cao et al. [98] concluded that there was no significant difference in symptom improvement and death rate in patients with severe disease between a group treated with Lpv/Rtv and another receiving a “standard” treatment. This suggests that Lpv/Rtv treatment is only effective when given at an early stage of the disease and in combination with other molecules such as Arbidol.
Remdesivir, an adenosine analogue with a broad antiviral spectrum, has been used to treat COVID-19 in different countries around the world but with controversial outcomes. Indeed, a multi-center study including 53 severe patients from three countries, 30 of whom required mechanical ventilation, reported that 68% of patients treated with remdesivir saw their condition improve after 18 days of hospitalization with a mortality rate of 13% [99]. Conversely, a randomized clinical trial conducted in China on 237 patients with severe forms of COVID-19 did not show a significant difference between the group treated with remdesivir and the control group [100]. However, another randomized clinical trial conducted in the United States on more than 1,000 COVID-19 patients, demonstrated better efficacy, since the administration of remdesivir significantly reduced the time of hospitalization and the mortality rate [101].
CQ is the controversial “molecule” that is still in debate today. In fact, this molecule has proven its antiviral power in vitro against SARS-CoV-2 [102]. CQ may be a drug of choice since it has few side effects and is well known by the scientific community as it has been prescribed for 70 years to treat malaria, rheumatoid arthritis, and certain forms of lupus [103]. Some researchers recommended its use for any intracellular infection, whether viral, fungal or bacterial [104]. However, clinical trials are needed to prove its effectiveness against SARS-CoV-2. Despite the fact that CQ was effective in vitro against dengue fever, all the following clinical trials were unsuccessful as it failed to reduce the hospitalization time [105,106].
In the current global health crisis, Algeria, among other worldwide countries, has advocated the use of CQ as a first-line treatment for patients with a moderate or severe form of COVID-19 [107]. This decision was motivated by the result of a preliminary clinical trial conducted in China in which nearly 100 COVID-19 patients were subjected to CQ treatment across 10 hospital centers; this treatment significantly reduced the hospital length of stay and the intensity of symptoms compared to the control group [108]. In addition, a study conducted in France on 36 patients with COVID-19, of whom 20 received treatment with HCQ with or without azithromycin, and 16 received symptomatic treatment, showed that after 6 days all of the patients treated with the HCQ-azithromycin combination were cured compared to 57% of patients treated with HCQ alone and 12% of patients in the control group [109]. Another study conducted by the same team on more than 1,000 COVID-19 patients demonstrated remarkable effectiveness, as nearly 99% of patients were cured after 15 days of hospitalization, with a death rate of 0.75% [110]. However, clinicians’ opinions remain divided on this matter and the main criticisms against these last two studies are the limited sample size for the first study, and the absence of a control group for the second study. In addition, the results of the British clinical trial “Recovery”, which is the first large and controlled randomized clinical trial are disappointing; indeed, HCQ did not reduce the time of hospitalization nor the mortality rate in COVID-19 patients during the 28-days hospital stay compared to the control group [111].
Facing an important variability in the symptoms and in the severity of SARS-CoV-2 infection, health practitioners are challenged to shorten the hospital stay or to save the patients’ lives by using all known and available therapies. Indeed, some studies suggest the use of glucocorticoids (GCs), while others prefer immunotherapy, including anti–IL-6 monoclonal antibodies and the serum of convalescent patients [112] to modulate the excessive or insufficient immune response. Moreover, others suggest the use of bronchodilators for the treatment of acute respiratory distress in critically ill COVID-19 patients [113].
Apart from the Lpv/Rtv combination that presents an increased risk of adrenal insufficiency due to the simultaneous use of inhaled GCs and Ritonavir [114,115], studies do not show any particular adverse effects of these treatments on COVID-19 patients with asthma or COPD. In addition, there are currently no specific guidelines on the therapies to use for COVID-19 patients with chronic respiratory comorbidities, but their management during this pandemic was the subject of several recommendations from public health organizations [116].
Asthmatics and COPD patients receive a basic treatment to control their disease in which inhaled or systemic GCs were used to reduce inflammation and bronchial hyperresponsiveness. However, administration of GCs at high doses (>150 mg/day) would increase the mortality rate in COVID-19 patients [52], but at lower doses studies have shown no difference in morbidity and mortality from COVID-19 between patients receiving GCs and control patients [117,118]. Other studies have suggested that they may even have a protective effect since prolonged use of GCs negatively regulates the expression of ACE2 [33]; moreover, ciclesonide, one of the GCs used in the treatment of chronic respiratory diseases, was able to inhibit SARS-CoV-2 replication in vitro [119].
Another study conducted with three COVID-19 patients over the age of 65, two of whom required oxygen therapy, showed encouraging results since the administration of this GC by inhalation improved the general health condition of the patients and significantly reduced viral load [120]. It is therefore recommended that asthmatics and COPD patients should continue their usual treatments with inhaled GCs in order to better control their disease, as this would allow them to reduce the frequency of exacerbations in the event of a viral infection with SARS-CoV-2 [121].
Additionally, special attention should be paid to the treatment of comorbidities, including allergic rhinitis and cardiovascular disease, as well as the management of exacerbations in asthma and COPD during the pandemic. In fact, allergic rhinitis is reported in over 78% of asthmatics and is considered to be a risk factor for the onset and exacerbation of asthma [122]. Moreover, the lack of control of allergic rhinitis is correlated with that of asthma [123]. Therefore, monitoring the treatment for allergic rhinitis at prescribed doses is strongly recommended, in parallel with that of asthma, specifically GCs administered by the nasal route [124] or allergen immunotherapy. Hence, for any new prescription of the latter, the sublingual method should be preferred to subcutaneous injection to avoid repeated medical visits [125].
Cardiovascular disease, including high blood pressure, is a very common comorbid condition in COPD patients, with prevalence rates up to 70% [126]. In addition, treatments used for cardiovascular disease do not have any adverse effects on COPD patients; they may even have some benefit in controlling the disease [127]. Consequently, the French and European hypertension societies recommend continuing the treatments based on the inhibitors of the renin-angiotensin system and antagonists of angiotensin receptors during the COVID-19 pandemic [128].
Finally, regarding the exacerbation of asthma and COPD, it would seem imperative to ensure that the patient masters the technique of inhaled bronchodilators to avoid hospitalization during this pandemic [129].
Conclusion
Improving current knowledge and understanding the impact of SARS-CoV-2 infection on respiratory diseases will help to improve the management of COVID-19 patients with chronic respiratory diseases. The prevalence of those comorbidities in COVID-19 patients differs between Chinese studies and European and American studies. Indeed, the reported rates of COPD and asthma in COVID-19 patients are significantly lower in China compared to the United States or Europe.
The impact of viral infections on chronic respiratory disease is well known; however, that of SARS-COV-2 remains controversial and may be underestimated or not fully understood. In addition, it appears that patients with respiratory diseases, including asthma and COPD, would not react in the same way to COVID-19. Indeed, infection with SARS-CoV-2 causes pneumonia and produces an imbalance in the ACE/ACE2 ratio and some biological parameters known to have negative effects on asthmatics and COPD patients that could worsen their symptoms and may increase their risk of exacerbation.
There is currently no specific treatment for COVID-19, but clinical trials of existing drugs are being carried out around the world. In this context, special attention should be paid to asthmatics and COPD patients’ treatments to avoid any risk of undesirable drug interactions, treatment interruption or overdose.
In conclusion, the evidence we have supports the idea that COVID-19 could have a more pronounced impact on asthmatics and COPD patients compared to those without comorbidities. However, further studies are needed to better elucidate the impact of this new infection on respiratory diseases and to better assess the potential risk for these patients.
Notes
Authors’ Contributions
Conceptualization: Bouazza B. Writing - original draft preparation: Bouazza B, Hadj-Said D. Writing - review and editing: Bouazza B, Pescatore KA, Chahed R. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
No funding to declare.



