Incidence and Risk Factors of Recurrent Venous Thromboembolism after Pulmonary Embolism
Article information
Abstract
Background
Information about the epidemiology of venous thromboembolism (VTE) recurrence in Korea is lacking. The purpose of this study was to investigate VTE cumulative recurrence rates and identify risk factors for VTE recurrence among Korean adults.
Methods
A retrospective cohort study was conducted on adult patients (≥18 years) admitted to a university teaching hospital for pulmonary embolism (PE) from 2005 to 2013. The main outcome of interest was a recurrence of VTE. We used Cox proportional hazard regression analyses to calculate the relative risk of VTE recurrence.
Results
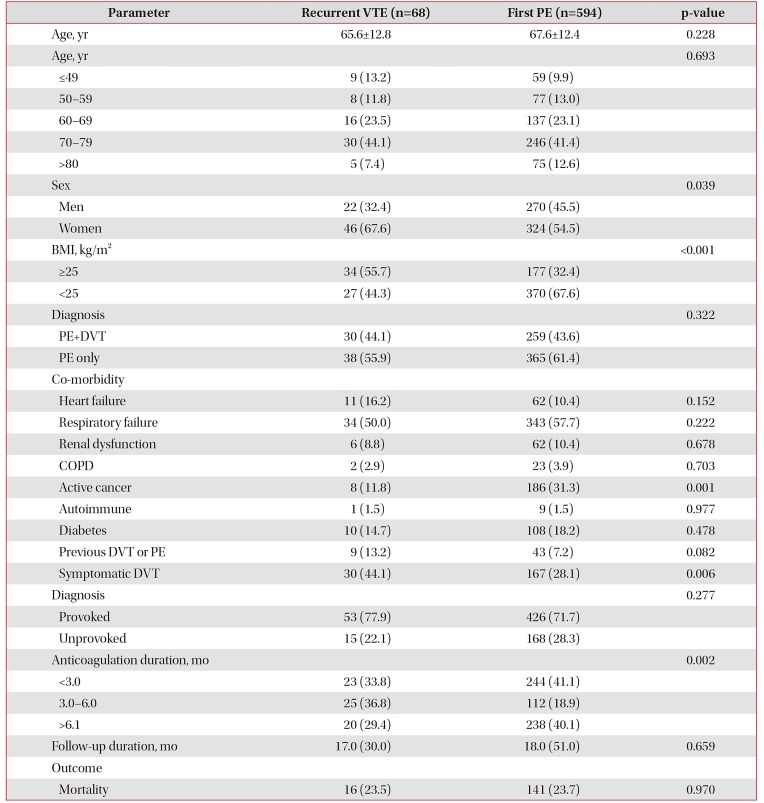
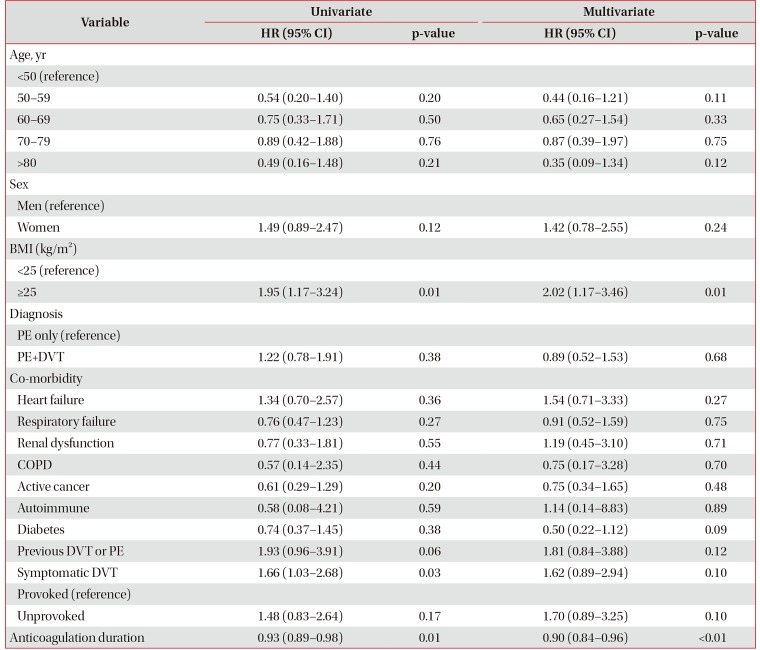
Five-year cumulative incidence of recurrent VTE events was 21.5% (95% confidence interval [CI], 17.7–25.4) in all cases of PE; 17% after provoked and 27% after unprovoked PE. Multivariate analysis showed that body mass index (BMI) of ≥25 (hazard ratio [HR], 2.02; 95% CI, 1.17–3.46; p=0.01) and longer anticoagulation therapy duration (HR, 0.90; 95% CI, 0.84–0.96; p<0.01) were independently associated with risk of VTE recurrence. Risk factors not found to be statistically significant at the <0.05 level included history of VTE (HR, 1.81; 95% CI, 0.84–3.88; p=0.12), unprovoked PE (HR, 1.70; 95% CI, 0.89–3.25; p=0.10), symptomatic deep vein thrombosis (HR, 1.62; 95% CI, 0.89–2.94; p=0.10), and female sex (HR, 1.42; 95% CI, 0.78–2.55; p=0.24). We found that age, history of cancer, and other co-morbidities did not significantly affect the risk of VTE recurrence.
Conclusion
Recurrence of VTE after PE is high. Patients with BMI ≥25 or reduced anticoagulation therapy duration have a higher risk of recurrent VTE.
Introduction
Venous thromboembolism (VTE) is a common, life-threatening disease that can frequently recur and cause long term complications. VTE comprises deep venous thrombosis (DVT) and/or pulmonary embolism (PE). The clinical presentation of about two-thirds of VTE cases is DVT, and the remaining one third present with PE1.
VTE is an important safety issue in patients who are on anticoagulation therapy. Early and long-term management is essential in order to prevent complications including recurrence of VTE, chronic thromboembolic pulmonary hypertension (CTEPH), and post thrombotic syndrome (PTS). The incidence of VTE in Asia is than in Western countries, but a recent retrospective study in the Korean population demonstrated an annual increasing incidence of VTE2.
In the western world, VTE is the third most common cardiovascular disease, following myocardial infarction and stroke34. Unlike in Western countries, the incidence of VTE in Korea is low, but it is increasing every year25. In addition, elderly women in Korea have almost the same incidence of VTE as that found in the West67.
Complications of VTE include PTS and CTEPH. PTS is a chronic venous insufficiency characterized by pain, swelling, and ulceration. It has been reported that PTS occurs in 20%–50% of patients with deep vein thrombosis, even after adequate treatment8. PTS is the most common complication of DVT, and is a risk factor for its recurrence91011. Incidence of CTEPH after PE is 1%–4%12131415. These chronic complications reduce quality of life and increase health care costs1617. VTE itself is economically burdensome, associated with significant increases in medical costs, especially those due to recurrence and hospital re-admission18.
There is a lack of research in Korea regarding the recurrence of VTE; therefore, our objective was to estimate the cumulative probability of recurrence of VTE after PE, as well as identify possible risk factors for these recurrences in Korea.
Materials and Methods
1. Study subject
From January 2005 to June 2013, 863 adult (PE) patients were consecutively enrolled in a single tertiary hospital, Keimyung University Dongsan Hospital, Daegu in Korea. The currentt study was approved by the institutional review board (IRB) at Keimyung University Dongsan Hospital, Keimyung University School of Medicine (IRB No 2015-06-045). The IRB waived the requirement for informed consent. This study was conducted in compliance with the Declaration of Helsinki. Of the 863 patients with pulmonary embolism, 201 were excluded because they had a follow-up period of less than one month. Recurrence of thromboembolic events was evaluated in the remaining 662 patients.
2. Follow-up and outcome classification
Electronic medical records of all PE patients were examined and all recurrent VTE events were noted. The median follow-up duration was 18.0 months (interquartile range [IQR], 0–49.0 months). Patients were treated with heparin and a vitamin K antagonist (VKA).
3. Statistical analysis
Baseline characteristics at diagnosis of PE (age, sex, body mass index [BMI], presence of risk factors, anticoagulation duration, and comorbidities) for both recurrent cases and non-recurrent cases were summarized using descriptive statistics (Table 1). A chi-square test was used to compare frequencies of risk factors between recurrent and non-recurrent cases. Cumulative recurrence of VTE was estimated using the Kaplan-Meier method. Cox proportional hazards modeling was performed to evaluate risk factors for recurrence. Second and third recurrent events after the index PE were not included in the cumulative incidence analyses. All statistical analyses were performed by using SPSS version 20.0 (IBM Corp., Armonk, NY, USA); p<0.05 was considered to be statistically significant.
Results
1. Incidence of recurrent VTE after PE
Of the total 662 patients with PE, recurrent VTE events were observed in 68 individuals during the follow-up period. Of these, 36 cases were of PE alone, 16 were of DVT alone, and 16 were of PE and DVT. Only one patient had recurrence of VTE during anticoagulation. Median follow-up period was 8.0 months (IQR, 3.0–22.25 months) for active cancer patients and 26.0 months (IQR, 8.0–60.0 months) for non-cancer patients. The 5-year cumulative incidence of recurrent venous thromboembolic events was 21.5% (95% confidence interval [CI], 17.7%–25.4%) after all PE. The 5-year cumulative recurrent venous thromboembolic rate were 17% after a provoked PE and 27% after an unprovoked PE (Figure 1).
2. Baseline characteristics
The baseline characteristics of the patients are provided in Table 1. Fifty-two patients had previous history of VTE. The majority of patients with provoked PE were females with a mean age of 65.6 years. Among the 68 patients with recurrent VTE, 53 patients had provoked PE and 15 patients had unprovoked PE. Women, and patients with BMI ≥25 kg/m2 and symptomatic DVT were higher among patients with recurrent VTE than in those without recurrent VTE. Anticoagulation duration was different between the two groups.
3. Risk of recurrent VTE
In the multivariate Cox regression analysis, patients with BMI ≥25 (HR, 2.02; 95% CI, 1.17–3.46) and longer anticoagulation therapy duration (HR, 0.90; 95% CI, 0.84–0.96) were significantly associated with risk of recurrent VTE during the follow-up period (Table 2). History of VTE (HR, 1.81; 95% CI, 0.84–3.88; p=0.10), symptomatic DVT (HR, 1.81; 95% CI, 0.84–3.88; p=0.12), unprovoked PE (HR, 1.70; 95% CI, 0.89–3.25; p=0.10), and female sex (HR, 1.42; 95% CI, 0.78–2.55; p=0.24) were associated with increased risk of a VTE recurrent event, but these factors did not reach statistical significance at the p≤0.05 level. Age, history of cancer, and other co-morbidities did not significantly affect the risk of a VTE recurrent event.
Discussion
There are four main findings from this study. (1) The cumulative recurrence rate of VTE was 17% in patients with provoked VTE and 27% in patients with unprovoked VTE 5 years after anticoagulation therapy. The rates of recurrent VTE found in this study are comparable to those in previous studies (15% and 30% respectively)1920. (2) Higher BMI (≥25) was associated with higher risk of recurrent VTE. (3) Longer anticoagulation therapy duration was significantly associated with reduced risk of recurrent VTE. (4) History of VTE, unprovoked PE, symptomatic DVT, and female sex were risk factors that trended toward an increased recurrent VTE rate, but they were not statistically significant.
There are a few reports in the literature which make it possible to compare recurrence rates of VTE in Asia with those in Western countries. Lee et al.21 report that the cumulative rate of VTE recurrence was 14.4% at 47 months (4-year) in a nationwide VTE study based on national health insurance in Taiwan. Nakamura et al.22 showed that in a warfarin treatment group, symptomatic recurrent VTE from time of randomization to an end point of either 12 months or study closure (overall study period) occurred in 24 of 538 of East Asian patients (4.5%) (including Korean, Japanese, Chinese, and Taiwanese) versus 122 of 3,854 non-East Asian patients (3.4%). In a recent meta-analysis, Yamashita et al.23 revealed that the absolute risk of recurrent VTE or VTE-related deaths was 3.3% and 2.6% with VKA in Asian and non-Asian patients respectively. These findings support the existence of a similar risk of recurrent VTE in Asians as in Caucasians and suggest that appropriate secondary prevention strategies for VTE should be emphasized in Asia.
In this study, a body mass index of 25 kg/m2 or greater (HR, 2.02; 95% CI, 1.17–3.46; p=0.01) was identified as a risk factor that significantly increased recurrent VTE. Importantly, a BMI of greater than 25 has been significantly associated with recurrent VTE in a dose dependent manner in other studies24. Since it has been shown that metabolic syndrome is a risk factor for VTE25, it may be necessary to control obesity to prevent VTE and its recurrence.
It is well known that the longer the duration of anticoagulation therapy, the less the risk of recurrence of VTE2627. In our study, the longer the duration of treatment, the lower the risk of recurrence of VTE, and the reduction found was similar to previous studies26. However, there are studies where prolonged anticoagulant treatment was shown to reduce the risk of recurrence, while the risk of relapse was not different from the start, when the anticoagulant was discontinued2829.
Active cancer is well known as a significant factor for associated with an increased risk for recurrent VTE303132. However, active cancer patients in the present study did not associated with recurrence of VTE. In this study, median follow-up period was 8.0 months (IQR, 3.0–22.25 months) for active cancer patients and 26.0 months (IQR, 8.0–60.0 months) for non-cancer patients. It was concluded that patients with active cancer died early and recurrent VTE was less due to low survival rate.
In this study, female patients (HR, 1.42; 95% CI, 0.78–2.55; p=0.24) showed a tendency towards an increased risk of recurrent VTE, although it has been previously reported that men have higher risk of recurrent VTE than women33. Interestingly, gender was not shown to be an independent predictor of recurrence in the Taiwanese study21. Thus, sex may not be a risk factor of recurrent VTE in Asian populations, unlike what has been found in Caucasian populations, but it will be important to explore this idea in well-designed prospective cohort studies.
In our study, history of VTE and unprovoked PE showed only a tendency to increase risk of recurrent VTE, with no statistical significance, although these two factors are considered to be associated with recurrence20. Because this study was performed retrospectively, missing data and mis-classification of associated factors could have been responsible for this discrepancy.
This study has some limitations that should be addressed. First, patients were enrolled from a single institute, retrospectively. Second, the oral anticoagulant agent used during the study period was a VKA. This was because the first available direct oral anticoagulant (DOAC), rivaroxaban, was not available before 2013 in Korea. Third, the time in therapeutic range of the international normalized ratio was not available for assessing the degree of quality control in the study. Fourth, antiphospholipid syndrome is an important risk factor for an increased risk for the first and recurrent VTE343536. However, in this study, the test for antiphospholipid antibody was rarely performed and excluded from the variables for risk factors.
We assessed the recurrence of VTE after anticoagulation therapy in patients with PE. in Korea. Our results warrant the conduct of a new prospective study to clarify the recurrence rates and to further develop a predictive model of recurrent VTE in the era of DOAC.
In conclusion, the recurrence rate of VTE after PE is high in Korea. Patients with higher BMI and shorter anticoagulation therapy duration have a higher risk of recurrent events.
Notes
Authors' Contributions:
Conceptualization: Hwang HG, Choi WI.
Methodology: Lee B, Lee CW.
Formal analysis: Lee B, Lee CW.
Data curation: Lee B, Lee CW.
Software: Lee B, Lee CW.
Investigation: Choi WI.
Writing - original draft preparation: Hwang HG, Choi WI.
Writing - review and editing: Hwang HG, Choi WI, Lee CW.
Approval of final manuscript: all authors.
Conflicts of Interest: No potential conflict of interest relevant to this article was reported.
Funding: This research was supported by the Soonchunhyang University Research Fund. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. This work was supported by the National Research Foundation of Korea (NRF) grant from the Korean Government (MSIP) (No. 2014R1A5A2010008).