 |
 |
| Tuberc Respir Dis > Volume 83(3); 2020 > Article |
|
Abstract
Background
Methods
Results
Notes
Authors’ Contributions
Conceptualization: Oh YM. Methodology: Oh YM. Formal analysis: Kim HK. Data curation: Lee H, Choi H, Kim SH, Lee JS, Lee SW, Lee JH. Software: Lee H, Choi H, Kim SH, Lee JS, Lee SW, Lee JH. Validation: Kim HK. Investigation: Kim HK. Writing - original draft preparation: Kim HK, Kim SH. Writing - review and editing: Lee H, Choi H, Lee JS, Lee SW, Lee JH, Oh YM. Approval of final manuscript: all authors.
Acknowledgments
Supplementary Material
Fig. 1.
Fig. 2.
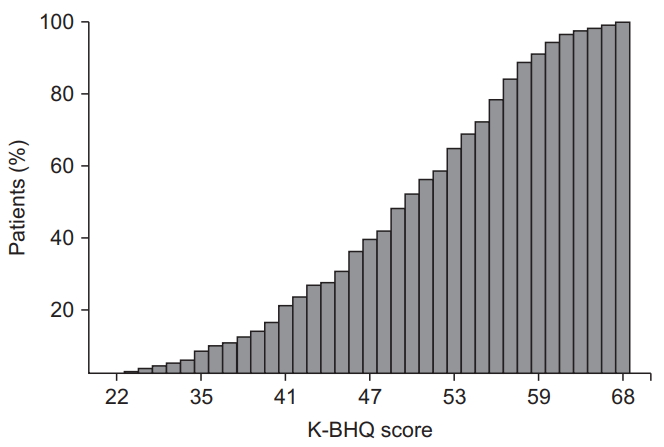
Fig. 3.
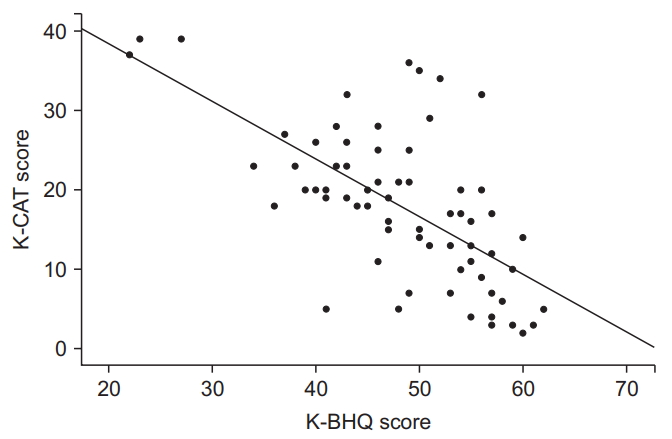
Fig. 4.
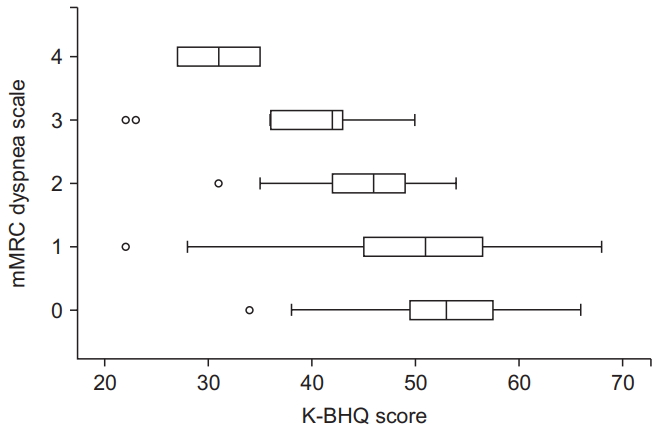
Fig. 5.
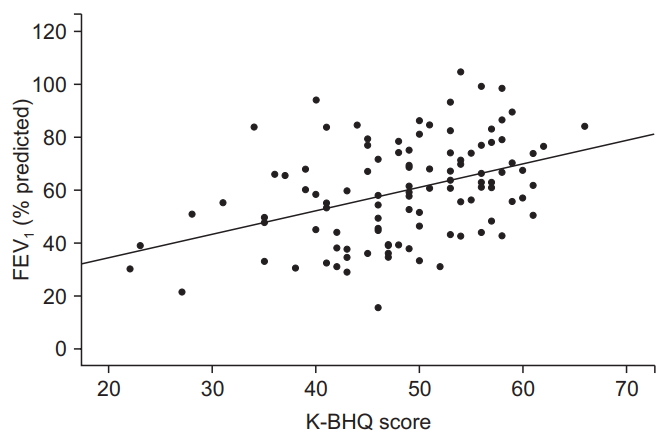
Fig. 6.
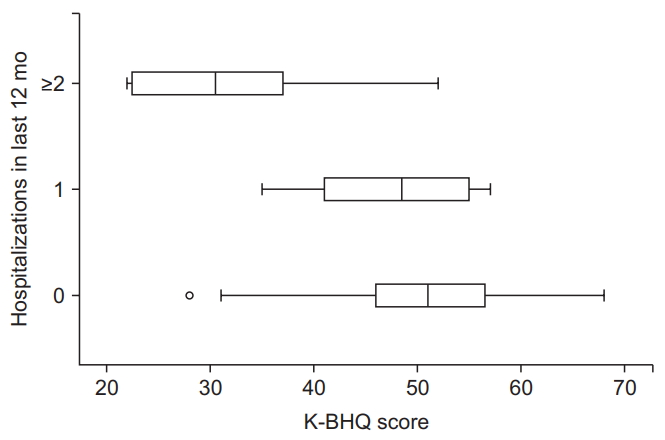
Table 1.
| Characteristic | Value (n=126) |
|---|---|
| Age, yr | 64.3±9.7 |
| Female sex | 67 (53.2) |
| BMI, kg/m2 | 22.7±3.4 |
| Smoking status | |
| Never | 75 (59.5) |
| Ex | 43 (34.1) |
| Current | 8 (6.3) |
| Previous pulmonary disease history | |
| Tuberculosis | 57 (45.2) |
| Measles | 30 (23.8) |
| Nontuberculous mycobacterium | 13 (10.3) |
| Pertussis | 13 (10.3) |
| Prebronchodilator spirometry (n=106) | |
| FEV1 % predicted (L) | 59.6±18.9 (1.62±0.60)* |
| FVC % predicted (L) | 71.7±16.6 (2.59±0.80)* |
| FEV1/FVC % | 54.4±25.5 |
| mMRC dyspnea scale | |
| 0 | 35 (28.8) |
| 1 | 65 (51.6) |
| 2 | 15 (11.9) |
| 3 | 9 (7.1) |
| 4 | 2 (1.6) |
| Exacerbations in past 12 mo† | |
| 0 | 82 (65.1) |
| 1 | 15 (11.9) |
| 2 | 15 (11.9) |
| 3 | 5 (4.0) |
| 4 | 5 (4.0) |
| 5 | 4 (3.2) |
| Exacerbations requiring hospitalization in last 12 mo | |
| 0 | 104 (82.5) |
| 1 | 14 (11.1) |
| 2 | 4 (3.2) |
| 3 | 3 (2.4) |
| 4 | 1 (0.8) |
| K-CAT score (n=70) | 17.6±9.6 |
| K-BHQ score (n=125) | 49.0±9.1 |
Table 2.
| K-BHQ correlation coefficient | p-value | |
|---|---|---|
| FEV1, % predicted | r=0.406 | <0.001 |
| FVC, % predicted | r=0.351 | <0.001 |
| Exacerbations in last 12 mo | ||
| Any | ρ=-0.245 | 0.006 |
| Mild to moderate* | ρ=-0.116 | 0.198 |
| Severe† | ρ=-0.303 | 0.001 |
| mMRC grade | ρ=-0.409 | <0.001 |
| K-CAT score | r=-0.656 | <0.001 |
References
- TOOLS
-
METRICS

- ORCID iDs
-
Hyun Kuk Kim

https://orcid.org/0000-0002-9360-3956Yeon-Mok Oh

https://orcid.org/0000-0003-0116-4683 - Funding Information
-
Korean Academy of Tuberculosis and Respiratory Diseases
- Related articles


 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Supplement
Supplement Print
Print Download Citation
Download Citation



