Introduction
Minute pulmonary meningothelial-like nodules (MPMNs) are small, benign, and often incidentally discovered in surgical or autopsy specimens of the lung. Since Korn et al.1 first described these small lesions in 1960, there have been many subsequent reports. Initially, MPMNs were thought to resemble chemodectomas. However, ultrastructural, and immunohistochemical findings suggest their similarity to meningiomas2. Some studies reported that MPMNs were likely reactive in nature3, and associated with pulmonary thromboembolism, respiratory bronchiolitis-associated interstitial lung disease, severe cardiac disease, and pulmonary adenocarcinoma2,4-6. The incidence of MPMNs varies from 0.3% to 9.5% at autopsy or surgical specimens2,3,5,7. There are just few MPMNs studies in Korea8,9. We report a case of MPMNs mimicking the appearance of hematogenous lung metastasis on a chest computed tomography (CT) scan.
Case Report
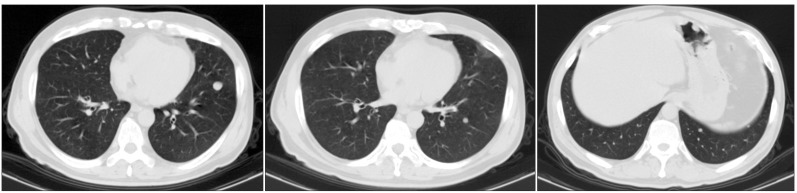
A 52-year-old man was referred to our clinic for an 11.3 mm nodule in the left lower lobe discovered on a chest CT scan. We found eleven small nodules of various sizes in both lungs, but there was no definite lymph node enlargement in the mediastinum or either hilum (Figure 1). The patient had complained of a persistent cough and mild dyspnea for several months. He had a history of secondary erythrocytosis, and was a current smoker with a history of 30 pack-years. He had no family history of cancer. His secondary erythrocytosis was mild, and he did not take any medication for it. His hemoglobin level was 18.2 g/dL, and the rest of the laboratory data revealed no abnormal results.
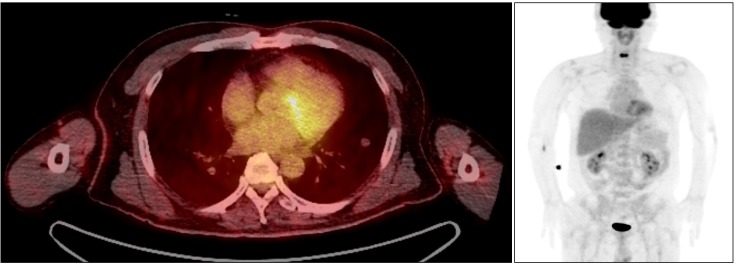
Initially, we decided to perform a biopsy, and consulted with the chest radiologist to perform a transthoracic needle aspiration using CT scan guidance. The pathologic report showed a few clusters of atypical cells that were suspicious for malignancy. However, cells acquired during needle aspiration were scant, so we could not make a definite diagnosis. For further evaluation, we carried out a positron emission tomography (PET) scan, esophagogastricduodenoscopy (EGD), and colonoscopy. There was no evidence of malignancy on the EGD or colonoscopy. The PET images showed multiple lung nodules scattered in both lungs. The largest one was 11.3 mm in the left lower lobe and did not have any discernible fludeoxyglucose uptake (Figure 2). We could not rule out the possibility of hematogenous lung metastasis from an unknown primary lesion.
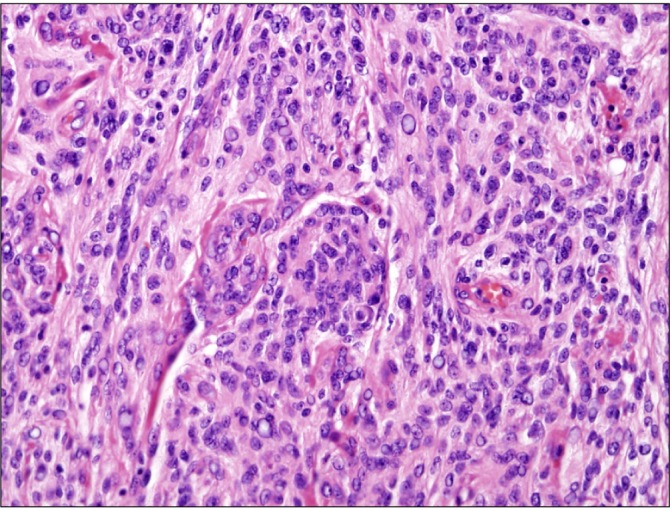
For pathologic confirmation, video-assisted thoracoscopic surgery (VATS) was performed while excising a lung parenchyma sample of 3.5├Ś1├Ś0.6 cm. Analysis revealed a relatively well-circumscribed gray-white solid mass, measuring 1.1 cm in diameter. Microscopically, the surgical lung biopsy specimen showed an interstitial proliferation of cytologically bland oval to blunted spindle cells. The cells enveloped the small blood vessels, and seemed to form whorls with dispersed nuclear chromatin (Figure 3). Immunohistochemical analysis was performed, and the sample was positive for epithelial membrane antigen (EMA), vimentin, CD56, and progesterone. Cytokeratin, thyroid transcription factor-1, and CD34 were negative. Finally, the diagnosis of MPMNs was established, and there was no evidence of malignancy. On the second postoperative day, the patient was discharged without any complications. Currently, he is being followed-up with at our hospital and is doing well.
Discussion
The origin of multiple pulmonary nodules may be benign or malignant, but multiple pulmonary nodules that are larger than 1 cm in diameter are most likely due to metastatic disease10. In this case, the patient presented with a 1.1 cm lung nodule in the left lower lobe on a chest CT, and we were initially suspicious of hematogenous lung metastasis. After VATS, the final pathologic report showed MPMNs. MPMNs are lung nodules often incidentally found in pulmonary parenchymal specimens.
When Korn et al.1 described MPMNs for the first time in 1960, they were thought to be benign proliferations of chemoreceptor cell precursors. Since then, there have been refutative studies showing that MPMNs have no endocrine granules, and are similar to meningothelial cells2,4. Therefore, the name of MPMNs was generally accepted in place of so-called pulmonary chemodectomas4. MPMNs are commonly asymptomatic, and their etiology is still unknown. According to one study, MPMNs are most frequently seen in the sixth decade, predominantly in women, and involve the upper lung lobes more often than lower lobes4. A study reported that MPMNs are found three times more often in the right lung than in the left2. The diameters of MPMNs range from 100 ┬Ąm to 3 mm. The prevalence of MPMNs is variable in each study, ranging from about 0.3-9.5% at autopsy or surgical resection2,3,5,7. In surgically resected specimens, the incidence increases up to 7-13.8%3,6. One study reported a 48% incidence rate in extensively-sampled lobectomies11. These differences in incidence rate might be caused by the study groups' variable main diseases.
Several studies suggested that MPMNs might be associated with chronic lung diseases such as pulmonary thromboembolism, respiratory bronchiolitis-associated interstitial lung disease, desquamative interstitial pneumonia, and atypical adenomatous hyperplasia2,5. Association with congestive heart failure was also suggested2. Chronic pulmonary diseases may stimulate the lung, and induce the formation of MPMNs, such as stretching or stiffening of the alveolar septa, or hypoxia, ischemia, parenchymal destruction, or some combination of these factors. The incidence rate of MPMNs has been reported to decrease in acute lung injury11. When we consider the results of previous studies, MPMNs are more likely to come from a reactive origin than a neoplastic origin. Also, there are no reports of cases in pediatric lungs, so we may exclude a congenital origin11. In one study about loss of heterozygosity, researchers found that isolated MPMNs lacked mutational damage, consistent with a reactive origin. However, multiple MPMNs show greater genetic instability, and more frequently have loss of heterozygosity. This suggests that MPMNs might represent the transition between a reactive and neoplastic proliferation7.
Although curative surgical excision is sometimes needed, MPMNs usually run a benign course, and conservative treatment is favorable. However, we should not overlook MPMNs, because some studies have described an association between MPMNs and lung cancer. Niho et al.3 reported that MPMNs were found in 10% patients with lung adenocarcinoma. In Mizutani al.'s study6, MPMNs were found more often in patients with malignant pulmonary tumors (especially lung adenocarcinoma) than in those with benign disease (7.3% vs. 2.5%, p=0.44) Although there is no guideline for follow-up in patients with MPMNs, we will follow the National Comprehensive Cancer Network guideline for lung cancer screening12 in this case. We will perform the low dose chest CT annually considering the risk of heavy smoker and history of MPMNs.
Immunohistochemical studies showed that MPMNs stained positive for EMA, vimentin, and progesterone and negative for cytokeratin, actin, CD34, S-100 protein, chromogranin, synaptophisyn, and neuron-specific enolase3,4,13. Our immunohistochemical staining results corresponded with those of previous studies. Niho et al.3 reported that half of MPMNs showed immunoreactivity for the progesterone receptor and suggests a role for sex-steroid hormones in the control of their growth. MPMNs in this case exhibited the immunoreactivity for the progesterone, therefore, close follow-up is needed for the growth of MPMNs.
In the present case, the sizes of MPMNs were larger than described in other studies. The pathological and immunohistochemical examinations provided a definite diagnosis of MPMNs. MPMNs may often be an incidental finding, and most cases do not require special treatment. However, when there is no confident clinical diagnosis, such as in our case, a pathological correlation is essential, despite the eventual diagnosis of MPMNs.
In summary, we report a case of MPMNs simulating the appearance of hematogenous lung metastasis on a chest CT scan. Minute pulmonary meningothelial-like nodules should be considered in the differential diagnosis of small lung nodules.


 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation






