 |
 |
| Tuberc Respir Dis > Volume 74(2); 2013 > Article |
|
Abstract
Background
Aiming to improve outcome of lung transplantation (LTx) patients, we reviewed risk factors and treatment practices for the LTx recipients who experienced respiratory infection in the late post-LTx period (>1 month after LTx).
Methods
We analyzed the clinical data of 48 recipients and donors from 61 LTx, who experienced late respiratory infections. Late respiratory infections were classified according to the etiology, time of occurrence, and frequency of donor-to-host transmission or colonization of the recipient prior to transplantation.
Results
During the period of observation, 42 episodes of respiratory infections occurred. The organisms most frequently involved were gram (-) bacteria: Acinetobacter baumannii (n=13, 31.0%), Pseudomonas aeruginosa (n=7, 16.7%), and Klebsiella pneumoniae (n=4, 10.0%). Among the 42 episodes recorded, 14 occurred in the late post-LTx period. These were bacterial (n=6, 42.9%), fungal (n=2, 14.3%), viral (n=4, 28.5%), and mycobacterial (n=2, 14.3%) infections. Of 6 bacterial infections, 2 were from multidrug-resistant (MDR) A. baumannii and one from each of MDR P. aeruginosa, extended spectrum β-lactamase (+) K. pneumoniae, methicillin-resistant Staphylococcus aureus and Streptococcus pneumoniae. Infection-related death occurred in 6 of the 14 episodes (43%).
Since the first human lung transplantation (LTx) in 1963, significant progress has been made in this field. From the registry of the International Society for Heart and Lung Transplantation (ISHLT), 3519 LTx for end-stage lung disease were performed in 2010. The median survival for LTx recipients, however, is 5.5 years, disappointing compared to that of other solid organ recipients1.
Morbidity and mortality throughout the post-LTx period result primarily from infection2-5; respiratory infections including pneumonia account for approximately 35% of deaths in the first year6. Among solid organ transplant recipients, LTx recipients are most susceptible to infection. Several factors unique to the lung may explain this. First, denervation of the allograft decreases mucociliary clearance and the cough reflex. Adding to the risks of generalized immunosuppression, the lung is the only allograft continuously exposed to the environment, donor-to-host transmission, and microorganisms colonizing the upper airways7.
The various etiologies of respiratory infection in LTx patients include opportunistic, hospital- and community-acquired microorganisms, which differ during the time to occurrence. Donor-to-host transmission is one of important risk factors in early respiratory infection following LTx7.
In the late post-LTx period (>1 month after LTx), the incidence of infectious episodes decreases markedly, despite patients returning to their normal activities at home or work. However, in this late phase, respiratory infection still presents a potentially fatal risk. Better understanding of post-transplantation susceptibility and of patterns of infectious exposure in the patient's environment is urgently required to avert this risk.
The aim of this study is to evaluate the epidemiology of respiratory infection in LTx recipients at our center. In particular, we analyzed the time to occurrence of infection, colonization of the recipient with a relevant infectious agent, donor-to-host transmission of these agents, and the relationship of late-phase respiratory infection to mortality.
We analyzed the medical records of 48 LTx recipients treated at our institution between January 2006 and June 2012 for demographic data, primary respiratory disease, microbiological testing before and after transplantation, and episodes of infection throughout the post-transplantation observation period. This study was reviewed and approved by Institutional Review Board at Gangnam Severance Hospital, Yonsei University College of Medicine (IRB no. 3-2012-0223).
Before LTx, sputum samples were cultured for bacteria and fungi, and broncho-alveolar lavage (BAL) fluids from recipient was analyzed by fibrobronchoscopy. Pre-transplant evaluations also included serology for cytomegalovirus (CMV), Epstein-Barr virus, hepatitis A, B and C, herpes virus, and human immunodeficiency virus.
Postoperative antimicrobial prophylaxis was guided by culture from donor and recipient. For the patients without known colonization, antibacterial prophylaxis was given as either a single agent, piperacillin-tazobactam or a combination of ceftriaxone, isepamicin and metronidazole. Systemic antimicrobial drugs were administered for 7 days if surveillance culture from donor and recipient were both negative. If the cultures were positive, antibacterial prophylaxis was maintained for at least 2 weeks as indicated by the antibiogram. All patients underwent prophylaxis for fungi with fluconazole daily for one year and for Pneumocystis jirovecii infection with sulfamethoxazole-trimethoprim. Based on CMV serology, patients at high and moderate risk for CMV infection (recipient [-]/donor [+] and recipient [+]/donor [+ or -], respectively) were maintained prophylactically with intravenous ganciclovir for two weeks, followed by oral valganciclovir for up to 6 months or one year.
Methylprednisolone was used to induce immunosuppression and was followed by maintenance therapy with steroids, calcineurin inhibitor and an antimetabolite. Maintenance immunosuppressive drugs consisted of prednisone, tacrolimus and mycophenolate mofetil.
Post-transplant evaluation included pulmonary function tests, imaging, periodic sputum cultures, and CMV antigenemia testing. If respiratory infection or rejection was suspected, the patient underwent a chest computed tomography scan, fibrobronchoscopy with alveolar lavage for bacterial and fungal cultures, indirect immunofluorescence tests for viral and P. jirovecii infections and mycobacterial polymerase chain reaction, and transbronchial biopsies to assess rejection and infection. Diagnosis of respiratory infection was made if a patient met any of the following criteria: fever (body temperature ≥ 37.8℃), cough, dyspnea, purulent expectoration, chest X-ray showing a new infiltrate, or significant impairment in lung function.
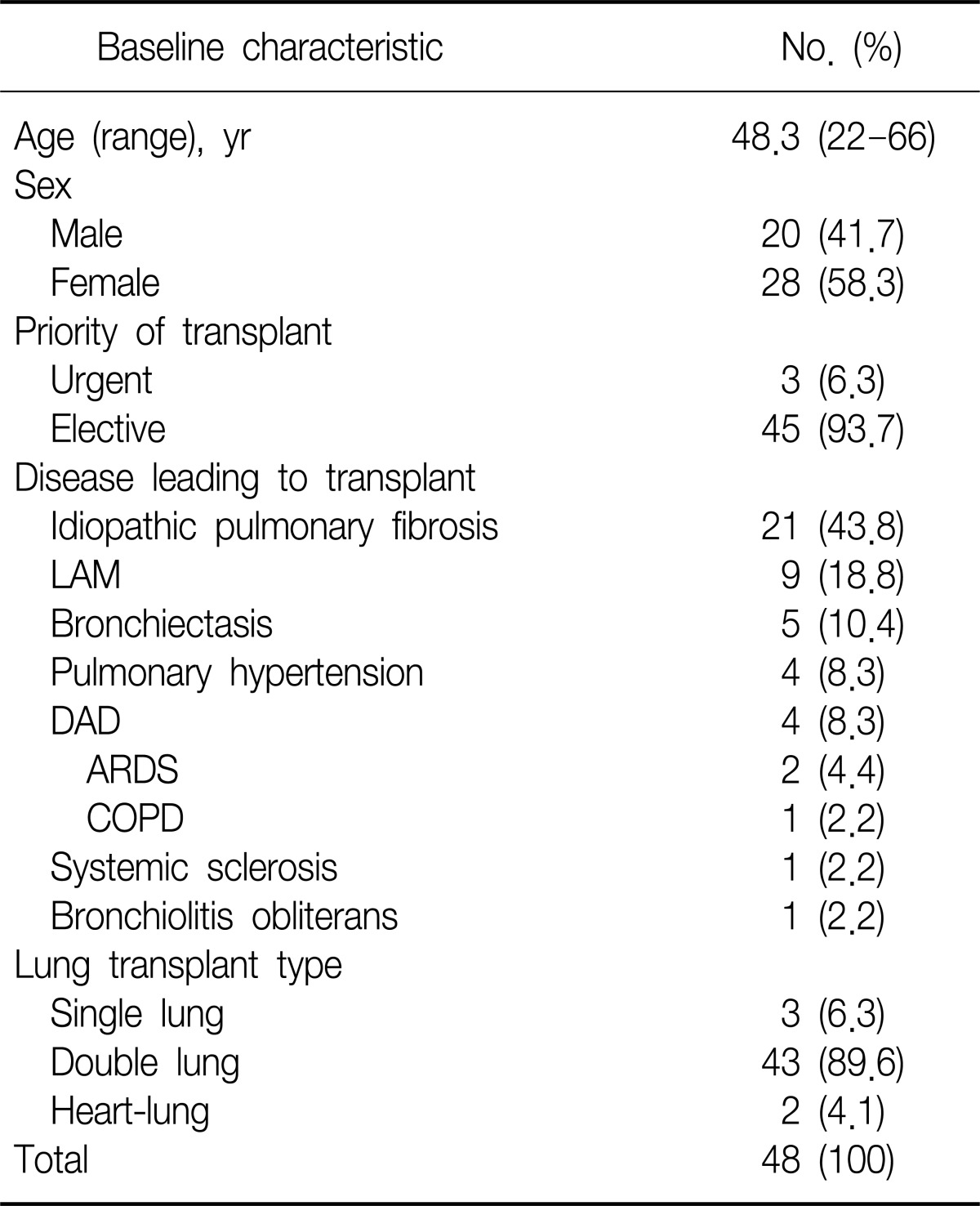
Demographic data for the LTx recipients are presented in Table 1. The mean age of the patients was 48.3 years. Idiopathic pulmonary fibrosis was the most common indication for LTx and most procedures were bilateral.
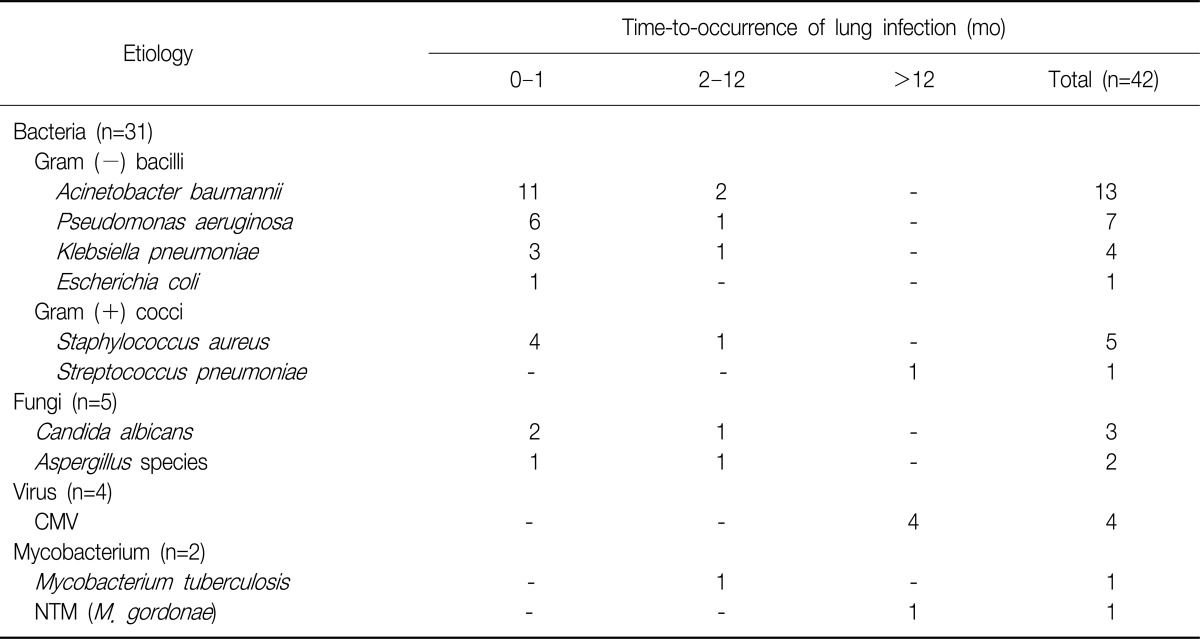
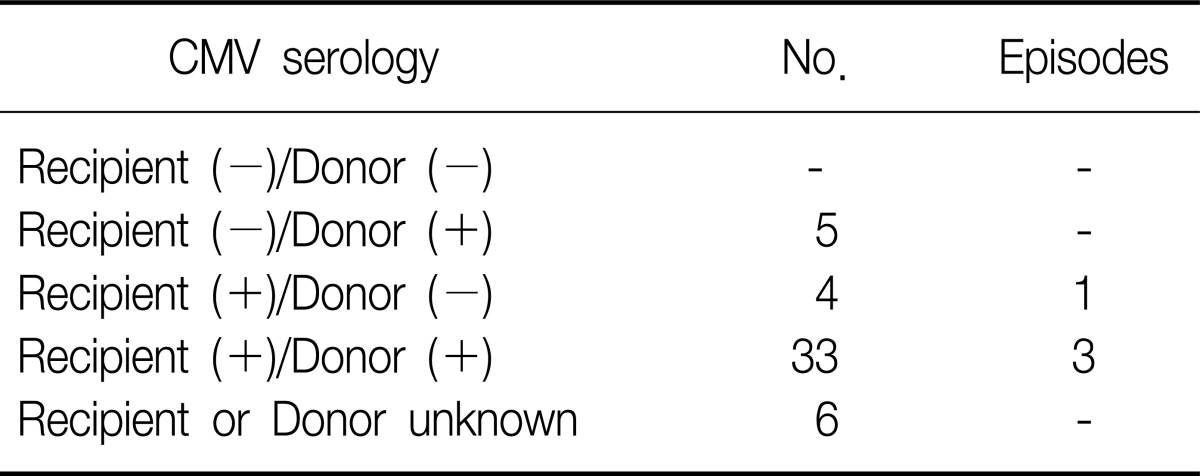
The microbiological etiology was established in 42 episodes from 48 recipient. Bacterial respiratory infection (73.8%) was more frequent than fungal (11.9%) and viral infection (9.5%). Bacteriological isolates were mostly (67.7%) gram (-) bacteria: A. baumannii (n=13, 31.0%), P. aeruginosa (n=7, 16.7%), and K. pneumoniae (n=4, 10.0%). Fungal infections were diagnosed in 5 recipients, all of them caused by either Candida albicans or Aspergillus species. Two cases of mycobacterial infection were identified, including one non-tuberculous mycobacterium (NTM): Mycobacterium gordonae (Table 2). All 4 patients with viral respiratory infection were diagnosed as CMV pneumonitis. However, no episode associated with CMV occurred within the first 12 months after transplantation. Reviewing the CMV serology, we found that none of the 5 high-risk mismatch recipients and 4 of the 37 medium-risk recipients developed CMV infection (Table 3).
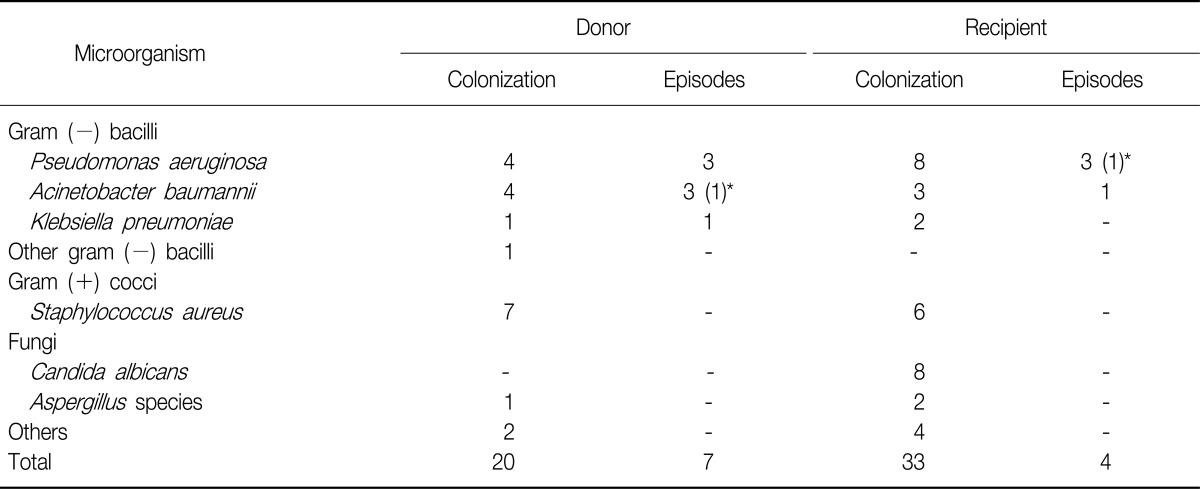
Among these, 7 patients (35%) were involved in donor-to-host transmission and 6 occurred within one month after LTx. Among 48 lung recipients in this study, 33 patients (68.8%) produced colonizing microorganisms in the pre-transplant BAL culture. Post-transplantation respiratory infection due to pre-transplant colonization occurred in 4 LTx recipients, and 3 of these infections occurred within the first month after LTx (Table 4).
Comparing most episodes (66.7%) of the respiratory infections occurred within the first month post-transplantation, 12 patients of the 48 LTx recipients had 14 episodes after one month. In contrast to episodes within the first month, in which bacterial infections predominated, late-phase infections showed diverse etiologies. These were bacterial (n=6, 42.9%), fungal (n=2, 14.3%), viral (n=4, 28.5%), and mycobacterial (n=2, 14.3%). Of 6 bacterial infections, 2 were from multidrug-resistant (MDR) A. baumannii and one each from MDR P. aeruginosa, extended spectrum β-lactamase (+) K. pneumoniae, methicillin-resistant S. aureus and S. pneumoniae. Among the 14 episodes of late-phase respiratory infection, 6 episodes (43%) resulted in death (Table 5).
The most critical risks for mortality in the first month after LTx from infection and posttransplant complications2,8,9. Risk of respiratory infection, typically by nosocomial organisms, is greatest in this early period. Prophylactic use of antibiotics reduced the risk of post-LTx pneumonia10. When immunosuppressive therapy is at the highest level, opportunistic organisms such as CMV and fungi account for most of the respiratory infections. Thereafter, community-acquired bacterial and viral infections also develop, although infection with health care-associated organisms remains common11.
In our analysis, the cumulative risk of an episode of respiratory infection increased sharply during the first 30 days after transplant, when more than half of the episodes occurred, and continued to increase until it stabilized. Bacterial pneumonia accounted for the largest proportion of infections followed by viral, fungal, and mycobacterial infections in the late period. Remarkably, most of the bacterial infections were gram (-) MDR and of nosocomial origin. These organisms also accounted for most of the respiratory infection-related deaths. Thus respiratory infections induced by gram (-) MDR organisms may predominate over community-acquired infection in the late period. Empiric antibiotic therapy in the early period after LTx is essential.
CMV infection ranks second to bacterial pneumonia in incidence in the first year following LTx12,13; however, our data revealed no episode of CMV in this early period. In accordance with previous studies, we attribute this relatively low incidence of early CMV infection and delay in onset time to the routine prophylactic use of ganciclovir for up to 6 months or one year14,15. Recent studies report a reduction in CMV viremia and infection with prolonged prophylactic regimens; however, the extended prophylaxis is more likely to delay the onset of CMV infection than to prevent it16-19. Other viruses such as herpes simplex, influenza, and respiratory syncytial viruses may cause pneumonia in LTx recipients; however, these did not appear in our study. In addition, prophylaxis with fluconazole at our institution proved effective in suppressing fungal infections. Previous studies show similar suppression of fungal infections as a result of prophylactic strategies20-23.
Other opportunistic microorganisms are far less common. Mycobacterial infections occur rarely after LTx and are typically secondary to Mycobacterium tuberculosis24. However, recent data show a rising incidence of NTM infections, particularly by M. abscessus, in 3% to 9% of cases25,26. Most previous data on pulmonary tuberculosis come from case reports or small series of patients and show incidence rates lower than 3%27,28. In our data, 2 mycobacterium infections occurred in 48 LTx recipients. These results agree closely with findings in other countries24-28, where the incidence of tuberculosis is much lower than in our country (80.7 episodes per 100,000 inhabitants/yr in 2011)29.
Data from ISHLT registry show that presence of clinical infection in a donor does not significantly predict mortality in the recipient in the first year30. Previous studies including ours confirm the presence of organisms in 60% to 80% of donor lungs, although these organisms very infrequently result in respiratory infections, especially in the late post-LTx period31-33.
This study is limited primarily in the short median follow-up period (338 days), small sample size and retrospective design. Additional observation and analysis of trends in respiratory infection after the first post-LTx year are needed. Meanwhile, prophylactic strategies currently in place should be continued.
In conclusion, although antibiotic therapy has sharply curbed the incidence of respiratory infections following LTx, respiratory infection still presents a major risk for mortality in the late post-LTx period. Nosocomial gram (-) MDR bacteria were most commonly involved in these episodes.
Acknowledgements
This study was supported by the institutional grant from Yonsei University College of Medicine (6-2011-0204) given to YS Chang.
References
1. Christie JD, Edwards LB, Kucheryavaya AY, Benden C, Dobbels F, Kirk R, et al. The Registry of the International Society for Heart and Lung Transplantation: twenty-eighth adult lung and heart-lung transplant report 2011. J Heart Lung Transplant 2011;30:1104-1122. PMID: 21962018.


2. Horvath J, Dummer S, Loyd J, Walker B, Merrill WH, Frist WH. Infection in the transplanted and native lung after single lung transplantation. Chest 1993;104:681-685. PMID: 8365275.


3. Maurer JR, Tullis DE, Grossman RF, Vellend H, Winton TL, Patterson GA. Infectious complications following isolated lung transplantation. Chest 1992;101:1056-1059. PMID: 1555420.


4. Paradis IL, Williams P. Infection after lung transplantation. Semin Respir Infect 1993;8:207-215. PMID: 8016481.


5. Chaparro C, Maurer JR, Chamberlain D, De Hoyos A, Winton T, Westney G, et al. Causes of death in lung transplant recipients. J Heart Lung Transplant 1994;13:758-766. PMID: 7803415.

6. Aguilar-Guisado M, Givalda J, Ussetti P, Ramos A, Morales P, Blanes M, et al. Pneumonia after lung transplantation in the RESITRA Cohort: a multicenter prospective study. Am J Transplant 2007;7:1989-1996. PMID: 17617864.


7. Campos S, Caramori M, Teixeira R, Afonso J Jr, Carraro R, Strabelli T, et al. Bacterial and fungal pneumonias after lung transplantation. Transplant Proc 2008;40:822-824. PMID: 18455028.


8. Dummer JS, Montero CG, Griffith BP, Hardesty RL, Paradis IL, Ho M. Infections in heart-lung transplant recipients. Transplantation 1986;41:725-729. PMID: 3012834.


9. Deusch E, End A, Grimm M, Graninger W, Klepetko W, Wolner E. Early bacterial infections in lung transplant recipients. Chest 1993;104:1412-1416. PMID: 8222797.


10. Husain S, Chan KM, Palmer SM, Hadjiliadis D, Humar A, McCurry KR, et al. Bacteremia in lung transplant recipients in the current era. Am J Transplant 2006;6:3000-3007. PMID: 17294526.


11. Burguete SR, Maselli DJ, Fernandez JF, Levine SM. Lung transplant infection. Respirology 2013;18:22-38. PMID: 22591266.


12. Zamora MR. Cytomegalovirus and lung transplantation. Am J Transplant 2004;4:1219-1226. PMID: 15268722.


13. Duncan AJ, Dummer JS, Paradis IL, Dauber JH, Yousem SA, Zenati MA, et al. Cytomegalovirus infection and survival in lung transplant recipients. J Heart Lung Transplant 1991;10(5 Pt 1):638-644. PMID: 1659899.

14. Soghikian MV, Valentine VG, Berry GJ, Patel HR, Robbins RC, Theodore J. Impact of ganciclovir prophylaxis on heart-lung and lung transplant recipients. J Heart Lung Transplant 1996;15:881-887. PMID: 8889983.

15. Zuk DM, Humar A, Weinkauf JG, Lien DC, Nador RG, Kumar D. An international survey of cytomegalovirus management practices in lung transplantation. Transplantation 2010;90:672-676. PMID: 20581765.


16. Ruttmann E, Geltner C, Bucher B, Ulmer H, Hofer D, Hangler HB, et al. Combined CMV prophylaxis improves outcome and reduces the risk for bronchiolitis obliterans syndrome (BOS) after lung transplantation. Transplantation 2006;81:1415-1420. PMID: 16732179.


17. Chmiel C, Speich R, Hofer M, Michel D, Mertens T, Weder W, et al. Ganciclovir/valganciclovir prophylaxis decreases cytomegalovirus-related events and bronchiolitis obliterans syndrome after lung transplantation. Clin Infect Dis 2008;46:831-839. PMID: 18269330.



18. Zamora MR, Nicolls MR, Hodges TN, Marquesen J, Astor T, Grazia T, et al. Following universal prophylaxis with intravenous ganciclovir and cytomegalovirus immune globulin, valganciclovir is safe and effective for prevention of CMV infection following lung transplantation. Am J Transplant 2004;4:1635-1642. PMID: 15367218.


19. Jaksch P, Zweytick B, Kerschner H, Hoda AM, Keplinger M, Lang G, et al. Cytomegalovirus prevention in high-risk lung transplant recipients: comparison of 3- vs 12-month valganciclovir therapy. J Heart Lung Transplant 2009;28:670-675. PMID: 19560694.


20. Hamacher J, Spiliopoulos A, Kurt AM, Nicod LP. Geneva Lung Transplantation Group. Pre-emptive therapy with azoles in lung transplant patients. Eur Respir J 1999;13:180-186. PMID: 10836345.


21. Reichenspurner H, Gamberg P, Nitschke M, Valantine H, Hunt S, Oyer PE, et al. Significant reduction in the number of fungal infections after lung-, heart-lung, and heart transplantation using aerosolized amphotericin B prophylaxis. Transplant Proc 1997;29:627-628. PMID: 9123449.


22. Calvo V, Borro JM, Morales P, Morcillo A, Vicente R, Tarrazona V, et al. Valencia Lung Transplant Group. Antifungal prophylaxis during the early postoperative period of lung transplantation. Chest 1999;115:1301-1304. PMID: 10334143.


23. Hosseini-Moghaddam SM, Husain S. Fungi and molds following lung transplantation. Semin Respir Crit Care Med 2010;31:222-233. PMID: 20354934.



24. Dromer C, Nashef SA, Velly JF, Martigne C, Couraud L. Tuberculosis in transplanted lungs. J Heart Lung Transplant 1993;12(6 Pt 1):924-927. PMID: 8312316.

25. Malouf MA, Glanville AR. The spectrum of mycobacterial infection after lung transplantation. Am J Respir Crit Care Med 1999;160(5 Pt 1):1611-1616. PMID: 10556129.


26. Chalermskulrat W, Sood N, Neuringer IP, Hecker TM, Chang L, Rivera MP, et al. Non-tuberculous mycobacteria in end stage cystic fibrosis: implications for lung transplantation. Thorax 2006;61:507-513. PMID: 16601086.



27. Paciocco G, Martinez FJ, Kazerooni EA, Bossone E, Lynch JP 3rd. Tuberculous pneumonia complicating lung transplantation: case report and review of the literature. Monaldi Arch Chest Dis 2000;55:117-121. PMID: 10949871.

28. Morales P, Briones A, Torres JJ, Sole A, Perez D, Pastor A. Pulmonary tuberculosis in lung and heart-lung transplantation: fifteen years of experience in a single center in Spain. Transplant Proc 2005;37:4050-4055. PMID: 16386624.


29. Korea Centers for Disease Control and Prevention. 2011 Annual report on the notified tuberculosis patients in Korea. 2011. Seoul: Korea Centers for Disease Control and Prevention.
30. Trulock EP, Edwards LB, Taylor DO, Boucek MM, Keck BM, Hertz MI. The Registry of the International Society for Heart and Lung Transplantation: twenty-first official adult lung and heart-lung transplant report 2004. J Heart Lung Transplant 2004;23:804-815. PMID: 15285066.


31. Zenati M, Dowling RD, Dummer JS, Paradis IL, Arena VC, Armitage JM, et al. Influence of the donor lung on development of early infections in lung transplant recipients. J Heart Transplant 1990;9:502-508. PMID: 2231088.

32. Weill D, Dey GC, Hicks RA, Young KR Jr, Zorn GL Jr, Kirklin JK, et al. A positive donor Gram stain does not predict outcome following lung transplantation. J Heart Lung Transplant 2002;21:555-558. PMID: 11983545.


33. Avlonitis VS, Krause A, Luzzi L, Powell H, Phillips JA, Corris PA, et al. Bacterial colonization of the donor lower airways is a predictor of poor outcome in lung transplantation. Eur J Cardiothorac Surg 2003;24:601-607. PMID: 14500081.



- TOOLS
-
METRICS

- Related articles
-
A Case of Acute Respiratory Failure Presenting Lobar Consolidation.1998 June;45(3)



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation



