Assessment of Household Catastrophic Total Cost of Tuberculosis and Its Determinants in Cairo: Prospective Cohort Study
Article information
Abstract
Background
One goal of the End tuberculosis (TB) Strategy is to see no TB-affected households experiencing catastrophic costs. Therefore, it is crucial for TB-elimination programs to identify catastrophic costs and their main drivers in order to establish appropriate health and social measures. This study aimed to measure the percent of catastrophic costs experienced by Egyptian TB patients and to identify its determinants.
Methods
We conducted a prospective cohort study with 151 Egyptian TB patients recruited from two chest dispensaries from the Cairo governate from May 2019 to May 2020. We used a validated World Health Organization TB patient-cost tool to collect data on patients’ demographic information, household income, and direct and indirect expense of seeking TB treatment. We considered catastrophic TB costs to be total costs exceeding 20% of the household’s annual income.
Results
Of the patients, 33% experienced catastrophic costs. The highest proportion of the total came in the pretreatment stage. Being the main breadwinner, experiencing job loss, selling property, and the occurrence of early coronavirus disease 2019 lockdown were independent determinants of the incidence of catastrophic costs. Borrowing money and selling property were the most-often reported coping strategies adopted.
Conclusion
Despite the availability of free TB care under the Egyptian National TB Program, nearly a third of the TB patients incurred catastrophic costs. Job loss and being the main breadwinner were among the significant predictors of catastrophic costs. Social protection mechanisms, including cash assistance and insurance coverage, are necessary to achieve the goal of the End TB Strategy.
Introduction
Tuberculosis (TB) is an infectious disease of major importance in public health and is among the top 10 causes of death globally, with 95% of its burden reported in low- and middle-income countries [1]. In addition to health-related burden (e.g., morbidity and mortality), TB poses a significant economic burden for patients, their households, and society. TB patients suffer from a substantial financial loss in direct out-of-pocket expenses for medical and non-medical costs incurred while seeking care [1]. In addition, each year, a TB patient loses an average of 3 to 4 months of work and up to 30% of household earnings [2,3]. One target of the End TB Strategy is that by 2030, no TB patient or household should face catastrophic costs due to TB [3,4]. Although there is no specific international metric to define catastrophic costs, TB-related direct and indirect income loss and out-of-pocket expenses exceeding 20% of the patient’s annual household income have been widely accepted as a threshold to classify TB patients experiencing catastrophic costs [5]. In 2016, the World Health Organization (WHO) adopted a definition that demarcates the threshold for catastrophic costs as 20% of a household’s total annual income in out-of-pocket and lost income costs due to TB [6,7]. Globally, it is estimated that TB patients and their households may experience catastrophic costs for health care that range from 27% to 71% [8,9]. Catastrophic costs reflect the economic burden of TB and barriers that make it hard for TB patients to access care. The magnitude of the costs and the coping strategies adopted by patients’ families in response can be a significant burden. This creates a barrier to diagnostic procedures and adherence to treatment, resulting in poor outcomes and financial suffering [4,10].
In Egypt, the National TB Control Program began to offer free TB-Directly Observed Therapy (DOTS) program to TB patients in the public sector in 2003 [11,12]. However, there currently are no social protection services for TB patients that help them cope with out-of-pocket and indirect costs during their treatment [12,13]. Currently, there is little evidence available about catastrophic costs experienced by Egyptian TB patients. To reduce barriers to access and adherence and to minimize the economic burden for TB-affected patients and their households, it is crucial that TB-elimination programs identify the main cost drivers, monitor financial hardship, and identify what further health and social policy measures should be taken [4,7,13].
To the best of our knowledge, programmatic data on the catastrophic costs incurred by Egyptian TB patients are not routinely collected, and few published studies are available on this topic. Because of the importance of the topic and its novelty in Egypt, we measured the incidence of catastrophic costs for TB patients in different phases of the treatment and identified its determinants among a sample of Egyptian patients.
Materials and Methods
1. Study setting and participants
We conducted the study at two chest dispensaries located in the Cairo governorate. These dispensaries provide TB treatment for most TB patients in Cairo. They do not serve sharply distinct patient populations (both serve patients with similar socioeconomic statuses living in the capital). We conducted the study from May 2019 to May 2020. Inclusion criteria included all newly diagnosed TB patients attending the studied dispensaries aged 18 years or more. We excluded any TB patient who was more than a month into TB treatment (for better recording of cost data in the pre-diagnostic period and for more timely following of the patients beginning from the start of their DOTS). We also excluded non-Egyptian TB patients (most of whom are refugees who receive substantial financial support from the United Nations monthly) and multi-drug resistant TB cases (because they require a much more intensive treatment regimen that lasts longer). We enrolled all patients who met the inclusion criteria consecutively by visiting the chest dispensaries three days per week until the required sample size had been reached at each site. We collected data by interviewing eligible TB patients four times (at the start of DOTS, after 2 months, after 4 months, and at the end of DOTS).
We calculated the needed sample size by assuming an incidence of catastrophic costs of 32% [10] with an 8% precision (25% of the expected incidence) to estimate a 95% confidence interval ranging between 24%–40%. We used a figure for catastrophic costs incidence from India to predict optimal sample size, because there were no available data about catastrophic costs in Egypt and because India’s socioeconomic standard is close to that of Egypt, both being lower-middle-income countries with similar rankings by the World Bank. An interim analysis conducted 3 months after the beginning of the study showed a rate close to the expected one.
The expected number of new cases in the selected centers used for the calculation of the sample size was 1,875 (from the official list of the NTP year 2019). Following these assumptions, we sought a sample size of 123 patients. To account for participants becoming lost to follow-up during the study, we inflated our sample size by 20% to reach a final target sample size of 154 patients. This sample size was sufficient for studying the determinants of the incidence of catastrophic TB costs, because we assumed a 20% difference in the incidence of such costs at an α level of 0.05 and a power of 80%. We calculated the sample by using open Epi program version 7.
The protocol was approved by the Research Ethical Committee of the Faculty of Medicine, Ain Shams University, Cairo, Egypt (No. FWA 000017585), done in accordance with the principles of the Declaration of Helsinki and operated under Federal Wide Assurance. We obtained written informed consent and obtained administrative approval for permission to conduct the study from the director of the National Tuberculosis Control Program and the included dispensaries’ managers.
2. Data collection tools
We adopted a standard interview questionnaire from WHO [4], with four sections, collecting the following information: socio-demographic characteristics; income-related data; time loss and total cost during the pre-diagnosis period; time loss, total cost, and coping strategies during and after TB treatment. We collected the data from the patients or their relatives. We used the same questionnaire during each of the four interviews (at the start of treatment, after 2 and 4 months, and at the end of the treatment). We calculated the costs incurred for every 2 months of treatment as total costs (less any donations or vouchers received) divided by total family income during this period. We calculated annual income as the sum of the amount of money received by the household in the year before the TB episode began, in exchange for labor or services, from the sale of property, as a profit from financial investments, or in the form of welfare payments [14]. We calculated the total cost during the period of DOTS from the sum of total medical costs, total travel costs, total food costs, total accommodation costs, and total income lost. If the total costs exceeded 20% of the total annual household’s pre-TB income, they were considered to be a catastrophic cost, as measured by dividing total cost by annual family income and multiplying the dividend by 100.
3. Definition of study variables
1) Catastrophic costs due to TB
Total costs (indirect and direct combined) exceeding a given threshold of the household’s annual income, here defined as exceeding 20%, as recommended by WHO [4].
2) Direct costs
Out-of-pocket payment for medical TB care (diagnostic tests, treatments, and follow-up tests) and non-medical costs (transportation, accommodation, and food/nutritional supplement costs) [4,13].
3) Indirect costs of seeking TB treatment
The productivity and economic costs of a patient or household incurred because of seeking TB health care and hospitalization during the TB episode. These we estimated by two methods: (1) self-reported household income loss less welfare payments and (2) total period of absence (in hours) multiplied by the hourly wage rate of the absent worker [4,13].
4) Poverty status
Those falling into the first quintile of household income per month were considered poor (i.e., ≤950 Egyptian pounds [EGP] per month). In this study, we used the international poverty line of $1.9 a day (US $57 per month) to compare the results with other studies that classify patients according to poverty status [13]. When we began the study, US $1 equaled 16 EGP; accordingly, US $57=946 EGP. The lowest quintile of the household monthly income of the study sample was 950 EGP; so we used 950 EGP per month as a cutoff point for classifying the study subjects’ poverty status.
4. Statistical analysis
We analyzed the data using SPSS statistical software version 24.0 for PC (IBM Corp., Armonk, NY, USA) and tested the data for normality using the K-S test. We described quantitative data as mean±standard deviations for normal data and as medians and interquartile range (IQRs) for non-normal data. Categorical data were presented as numbers and percentages. We described costs as median and IQRs in per-patient costs in US$ using the mean UN exchange rate for the US$ in 2019 (US $1=16 EGP). We calculated the proportion of households that experienced catastrophic costs and used a binary response model to identify the factors most significantly associated with catastrophic costs, using a χ2 test with two-tailed p<0.05 considered to be the threshold for statistical significance. We used multivariate logistic regression by the enter method to identify differences in the model’s fitness. The model used independent variables with p<0.2 to enter the model, i.e., sex, residency, crowding index, breadwinner, type of TB, loss of job, stopping education, selling property, and coronavirus disease (COVID) lockdown. We calculated both the crude and the adjusted odds ratios (aORs). The model predicted 82% of the responses correctly, and the value of the Nagelkerke R2 was 0.57.
Results
A total of 151 TB patients were contacted and consented to being included in the study, which was 98% of the initial goal sample size of 154. Three cases were excluded because of failure to complete the interview. Table 1 presents the basic socio-demographic and economic characteristics of the patients. Their ages ranged from 18 to 76 years, with a mean age of 39±15.7 years. More than half the patients (60.3%) were males. Married patients represented 55.6% of the sample, and 42.4% of the participants had completed secondary school or above. More than half the patients had jobs (50.3%), and 56.3% were the main breadwinners of their families. The median household size of the patients was 4 (with a range of 1 to 11). The median number of rooms in the home was three (with a range of two to five rooms). In 70% of the cases, the TB was pulmonary TB. The median (IQR) duration for pre-treatment from initial care-seeking to being diagnosed for TB was 90 days (range, 30–120). About two thirds of the participants (64.9%) began to receive their DOTS within 3 months of the start of symptoms.
Table 2 indicates that the median (IQR) of the total indirect cost equaled US $0.0 to US $281.3 and represented 70.5% of the total cost. The median (IQR) of the total direct cost was US $48.06 (29.3 to 96.9), representing 29.5% of the total cost. The highest median cost was for travel ($21.25), followed by the medical costs ($17.5), and the lowest median cost was for food ($1.25). As a percentage of total cost, medical costs had the highest share (11.6%), and food costs represented the lowest share (6.4%). In the pre-treatment phase, the median for the medical costs (US $15.63) was higher than that of the travel cost (US $5.63) or food cost ($0.0), and the percentages of the total cost showed the same trend. In the following three phases, both the median cost and the percentage for the travel cost was higher than those for medical and food costs. In general, the median of the total direct cost showed a decreasing trend during the following phases, ranging from US $24.38 in the pre-treatment phase to US $2.5 over the last 2 months. The table also shows that accommodation was not considered a burden among the study participants.
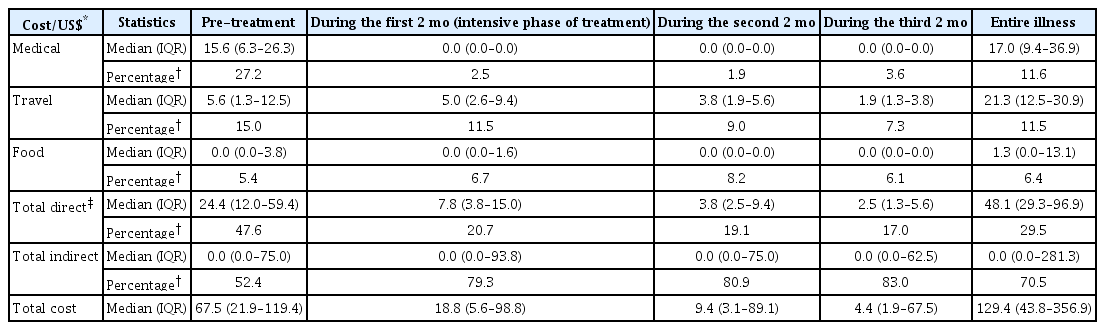
The estimated costs that are borne by TB patients’ households affected during the entire illness and during different stages
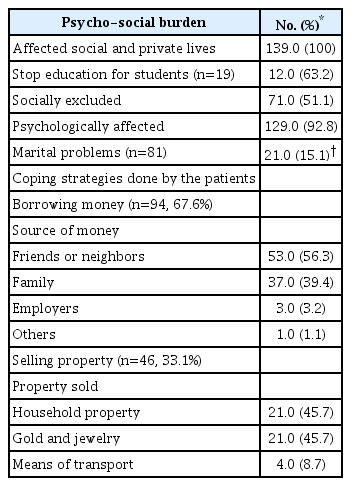
All of the patients who answered the prompt about the negative impact of TB reported that their social and private life had been affected. Most participants had experienced psychological problems (92.8%). Almost half the patients had been socially excluded (51.1%). Job loss was reported by about a third of the patients (31.6%), and marital problems were reported by 15.1% of them. Four patients became divorced during the study. To cope with the challenges, about two thirds of the patients borrowed money (67.6%), and about 33% of the patients sold property (Table 3).
In all, 33.1% of the study participants and their households experienced catastrophic costs (50/151). The incidence of catastrophic costs among males (49.5%) was significantly higher than that among females (8.3%) (χ2=27.6, p<0.001). The incidence of those experiencing catastrophic costs in the older group was higher (34.8%) than the 31.8% among the younger age group, but the difference was not statistically significant (χ2=0.4, p=0.527) (Table 4).
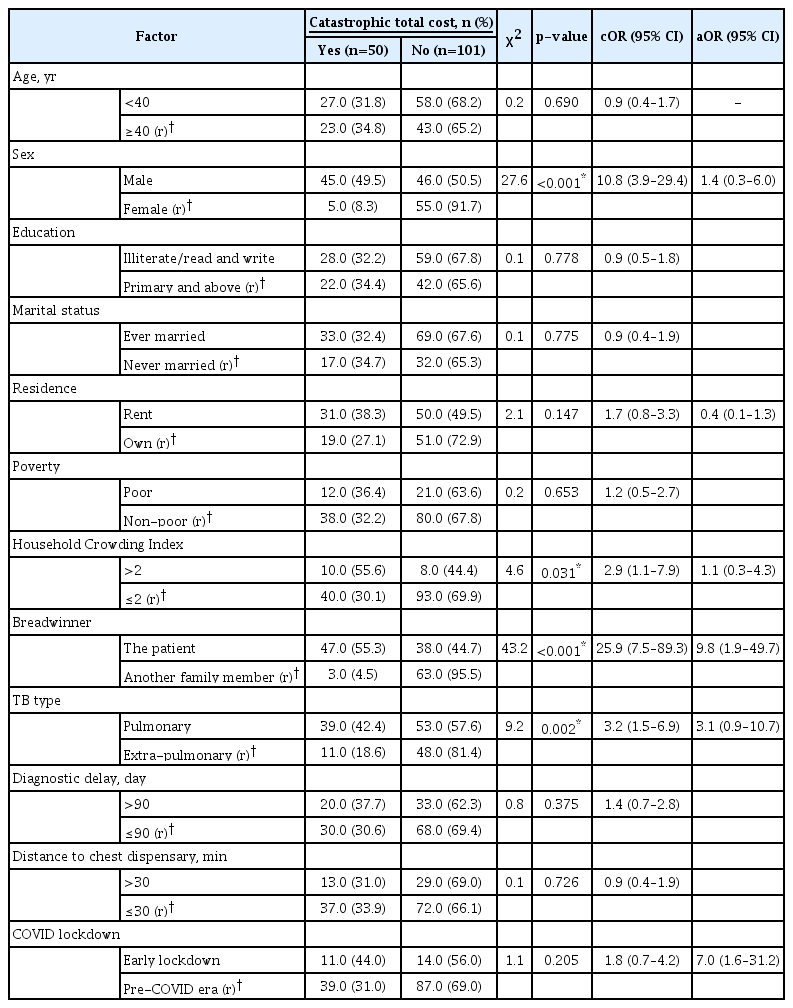
Association between catastrophic total cost and the patients’ personal and demographic characteristics (n=151)
Table 4 also shows which of the patients’ personal and demographic characteristics were the main drivers of catastrophic costs. Among them, there was a statistically significant association between the catastrophic total cost and sex (p<0.001), type of breadwinner (p≤0.001), crowdedness index (p=0.031), and type of TB (p=0.002). Loss of employment (p<0.001) and selling property (p=0.019) were also significantly associated with the catastrophic total cost, as is clear from Table 5. Among the independent factors with p<0.2 that were entered into the logistic model, the only significant predictors of catastrophic costs were being the family breadwinner (aOR, 9.76; 95% confidence interval [CI], 1.92–49.68), early COVID lockdown (aOR, 7.05; 95% CI, 1.59–31.24), job loss (aOR, 6.52; 95% CI, 2.15–19.84), and selling property (aOR, 4.74; 95% CI, 1.49–14.96). The log odds of the total catastrophic costs can be calculated from the following regression function:
(−4.686+2.278×breadwinner+1.555×selling property+1.876×loss of job+1.95×COVID lockdown)
We also did cost subgroup analyses for gender, type of tuberculosis, COVID-19 lockdown, and being the family breadwinner or other member (Supplementary Table S1).
Supplementary Tables S1 and S2 show that the median total cost among females was not significantly higher than that among males (mainly because of the significant difference in their median total direct cost), but at the same time, the median annual income was not significantly higher among females, which may compensate for the increase in total cost and explain why the percentage of the catastrophic total cost was higher among males than among females, as shown in Table 4.
The median total cost was higher among family members (US $182.19) who were not breadwinners than among those who were (US $106.88). This difference was not significant (p=0.054); but the median annual income, which was the denominator used for calculating the catastrophic total cost, was higher among those who were not family members, neutralizing the effect of the higher cost, which could also explain the higher percentage of the catastrophic total cost among non-breadwinners (Supplementary Tables S2, S3).
The median total patient costs were not significantly decreased after COVID 2019 (COVID-19) lockdown (Supplementary Tables S2, S4), apparently because the patients’ direct costs for seeking care were reduced after the lockdown period. However, the median annual income of post–COVID-19 TB patients was significantly lower because of the lockdown as well. This could help explain why the incidence of catastrophic cost increased for patients in the post–COVID-19 lockdown period.
For the type of TB, the median of all the costs was not significantly higher among pulmonary TB patients than among extra-pulmonary patients (Supplementary Tables S2, S5).
Discussion
This prospective cohort study assessed major health problems in Egypt (TB). To our knowledge, there have been no previous studies on catastrophic TB costs in Egypt. A strength of this study was that it was longitudinal and collected data by interviews at four discrete times during a 6-month DOTS treatment, allowing better and more accurate collection of cost data with less dependence on patients’ recollection. It did not prove feasible to interview patients each time they came to receive their DOTS; so we reduced the data collection to four interviews to improve compliance and increase the response rate. We assessed the profile and catastrophic impact of TB care costs on the households of TB patients enrolled at two chest dispensaries in the Cairo governorate.
In total, 33.1% of households experienced catastrophic costs due to TB care. This finding agrees with the results of a systematic review in low- and middle-income countries which reported that 39% of the patients studied faced catastrophic costs [15]. It is also near the range of findings of Asian studies conducted in India and Indonesia that have reported 32.4% and 36% of households, respectively, as having experienced catastrophic costs due to TB care [10,13]. Similarly, studies conducted in South Africa, Kenya, and Peru found that 28%, 26.1%, and 39% of participants faced catastrophic costs, respectively [7,8,16]. However, our results do not match the incidence rates reported by studies conducted in other regions. For instance, they were lower than the rates reported in studies conducted in China (52.8%) [17], in India (68%) [18], in Vietnam (63%) [9], in South Africa (55%) [19], and in Ethiopia (53.6%) [12]. This disagreement may result from the inclusion of TB patients with human immunodeficiency virus (HIV) in those studies, whereas only pulmonary TB cases were reviewed here; the use of a 10% threshold for annual income to define catastrophic cost; or the use of nationwide data as opposed to our local study. However, our results were much higher than those reported in the Kassa study in Benin, which found that 14.8% of TB patients experienced catastrophic costs [5]. Another study conducted in Delhi reported that only 7% of TB patients faced catastrophic costs [20], perhaps because those studies included children, and, unlike this study, were conducted prior to the COVID-19 pandemic, which had economic effects on most people globally and added more costs faced by TB patients.
About half the studied TB patients were employed (76/151); however, 57.9% (44/76) lost their employment during the illness period. This result is higher than that reported in a previous Indonesian study conducted in 2018 by Faudy et al., who found that 32% of the patients lost their jobs while undergoing treatment for TB [13]. Another study done in the Philippines (2020) indicated that about 46% of participants lost employment [20]. These differences in job loss may relate to differences in the socioeconomic status and type of job of the participants, because 25% of our study participants were working as street peddlers or doing informal paid work. In addition, 56.3% of our participants were the main breadwinners for their families. These two factors reflect the indirect economic burden of TB and how it may deprive a family of income throughout the duration of an illness. This also highlights how important it is for governments to develop and implement strategies that provide a source of income for families during the illnesses, especially when the patient is the only breadwinner in the household.
About two thirds of our participants started to receive their DOTS within 3 months of appearance of their symptoms, much as in several previous studies conducted in Uganda, Bangladesh, and Kenya [6,8,21]. In addition, our patients experienced the largest share of the total costs in the pre-treatment stage. This agrees with Asres et al. [2], Foster et al. [22], and Tanimura et al. [15], who reported that more than half the total costs in their studies were incurred during the pre-diagnosis period. This highlights the importance of mobilizing governmental resources toward earlier detection and support for TB patients, to reduce diagnostic delay and pre-treatment costs.
In this study, medical cost represented the highest percentage of the total direct cost in the pre-treatment stage. Direct costs for travel and food were higher than were the direct medical costs in the following stages, as in a study in 2014 done by Wingfield et al. [16], who reported that, during the pre-treatment stage, medical cost was the highest share (about two thirds) of the direct costs. This reflects the problem of diagnostic delay, which leads to patients’ remaining ill despite seeking medical care, frequently in the attempt to receive a proper diagnosis.
There was a statistically significant association between the incidence of catastrophic cost and certain personal, medical, and social characteristics, such male gender, being a breadwinner, crowded household, pulmonary type of TB, job loss, undergoing early COVID-19 lockdown, and sale of property. These factors and others were reported by other studies conducted in various countries [7,23,24]. This reflects that providing free coverage of only the anti-tuberculosis treatment to a patient is not enough to alleviate the catastrophic costs they face; identifying the independent determinants of catastrophic costs is crucial for tailoring prevention and control policies.
All of our patients reported that their social and private lives were harmed, and most of them experienced psychological problems and social exclusion. Many previous studies had also found that more than half the participants reported that TB profoundly affected their social lives and well-being [8,21,24]. For the coping strategies adopted by the patients, we found that about two thirds of the patients borrowed money and about a third sold property. This result is in keeping with a study conducted in Uganda [21] which revealed that >50% of the patients adopted a coping mechanism, such as borrowing money or selling assets. It also fits comfortably with Laokri et al. [24], who reported that 37% of the patients borrowed money and 53% sold household assets, and with another study which found that 38% of TB patients reported borrowing money. However, our figures are much higher than those in two South African studies; Foster et al. [22] found that only 5% of patients sold assets, and Mudzengi et al. [19] found that taking a loan was reported by only 8% of the patients, perhaps because in South Africa, disability grants are provided to patients who cannot work because of illness [22].
Catastrophic costs due to tuberculosis constitute a substantial economic burden and harm patients and their households. Thus, implementing comprehensive screening programs for early detection of TB and adding additional strategies to the National TB Control Program to not only freely cover the treatment but also target the problem of indirect costs and loss of income will be crucial in the prevention and control of this problem.
The limitations of a study are as follows. First, we did not incorporate the costs experienced by multi-drug-resistant TB patients, because they undergo a different treatment regimen, which last longer than 6 months. Our study followed patients throughout a course of treatment that lasted for only 6 months. We also excluded TB patients with HIV, because its prevalence is low in Egypt (<0.1%) [25]. The effect of hospitalization was also excluded, because we could not include hospitalized patients, whose omission likely led to underestimating the catastrophic costs experienced by TB patients.
Second, the health system imposes pre-diagnostic delays, the determinants of which we did not analyze in this study, since many TB patients are not covered by a health insurance system in Egypt, and the public-health system is currently in only the initial stages of reform to achieve universal health coverage. This limitation likely led to an underestimation of TB care costs and the level of catastrophic costs borne by TB patients.
Third, our study began in the pre–COVID-19 era and ended after the lockdown that began in March 2020. Because of this lockdown, most of the Egyptian population lost income, although the medical costs of some, who were afraid of getting an infection if they came to the chest dispensaries to receive their treatment, were decreased. Thus, the fact of this lockdown could have increased catastrophic costs. Although the inclusion of both pre– and post–COVID-19 TB patients in our study likely distorted the results, that may be a strength, because subgroup analysis provided important insights for future research on how COVID-19 and lockdown policies affected TB patient costs.
Fourth, using a convenience sample from two similar dispensaries to assess patient costs did not guarantee adequate representation of the entire population of TB sufferers in the Cairo governorate, thus limiting the generalizability of our findings.
Despite the provision of free TB care under the Egyptian National TB Program, nearly a third of the study participants incurred catastrophic costs, with an indirect cost greater than the direct one throughout the entire illness. Being the family breadwinner, experiencing job loss, selling property, and undergoing early COVID-19 lockdown were the most significant predictors of the catastrophic total cost.
Continuous nationwide monitoring of the incidence and determinants of catastrophic costs for a better definition of the TB patients’ needs is recommended. The provision of cash grants and vouchers to enable TB patients to obtain necessary nutritional and medical supplements at a reduced price is required if the goal of the End TB Strategy is to be achieved. Other issues, such as health-system delays, multidrug-resistant TB, and the effects of hospitalization, require further study.
Notes
Authors’ Contributions
Conceptualization: Ellaban MM, Basyoni NI, Boulos DNK, Rady M, Gadallah M. Methodology: Ellaban MM, Basyoni NI, Boulos DNK, Rady M, Gadallah M. Formal analysis: Ellaban MM, Basyoni NI, Boulos DNK, Rady M, Gadallah M. Validation: Rady M, Gadallah M. Investigation: Ellaban MM, Basyoni NI, Boulos DNK, Rady M, Gadallah M. Writing - original draft preparation: Ellaban MM, Basyoni NI, Boulos DNK, Rady M, Gadallah M. Writing - review and editing: Ellaban MM, Basyoni NI, Boulos DNK, Rady M, Gadallah M. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
No funding to declare.
Acknowledgements
We thank the NTP team for enabling this research and providing support. We are also grateful to all of the participating patients for their cooperation.
Supplementary Material
Supplementary material can be found in the journal homepage (http://www.e-trd.org).
The estimated cost (US$*) incurred by TB patients according to sex.
The difference in the median costs and annual income between different subgroups.
The estimated cost (US$*) incurred by TB patients according breading status.
The estimated cost (US$*) incurred by TB patients according to pre/post‒COVID-19 lockdown.
The estimated cost (US$*) incurred by TB patients according to the type of TB.