Confocal Laser Endomicroscopy in a Critically Ill COVID-19 Patient
Article information
Probe-based confocal laser endomicroscopy (pCLE) is a minimally invasive technique for in vivo real-time microscopic imaging of proximal and distal airways during bronchoscopy. Using the laser beam with a wavelength of 488 nm, it allows detection of autofluorescent alveolar, vascular, cellular, and noncellular microstructures with an optical area of 600 μm and a focus depth of 50 μm [1]. The role of pCLE has not been clearly established in patients with coronavirus disease 2019 (COVID-19), especially correlating with high-resolution computed tomography data, bronchoalveolar lavage, and traditional histopathology.
The patient gave written informed consent for pCLE on the day of admission to the clinic. The study protocol was approved by local ethics committee of clinical center.
An age 70 male who never smoked was admitted COVID-19 positive on the 3rd day of illness. The patient’s respiratory failure progressed rapidly. pCLE and high-resolution computed tomography were performed the next day after intubation. pCLE has been done in four right and three left lung segments (Figure 1). In alveolar spaces, pCLE showed large highly fluorescent floating structures 50×240 μm. Alveolar structure exhibited increased elastin fiber thickening, dystelectatic abnormalities. Disorganized microvessel growth was also observed.
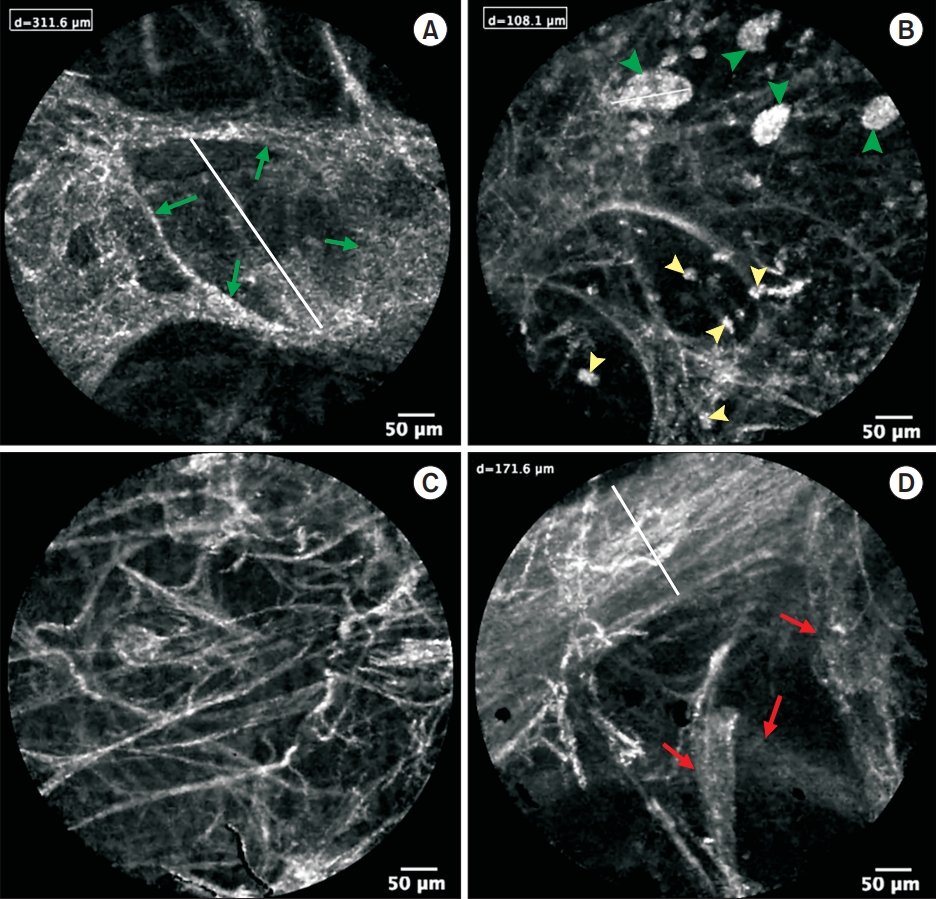
Endomicroscopic images of distal airways in active coronavirus disease 2019 patient (on the 9th day of hospital admission) show the loss of organization, increased connective tissue fibers and structures, which may correspond to areas of granulation tissue on histological image (B), only single alveoli (green arrows) are visualized (A). Highly fluorescent large floating structures (green arrowheads), alveolar macrophages (yellow arrowheads) (B) are present in the alveolar compartment. Signs of dystelectasis with an increase in the number of connective tissue structures in the field of view (C) and disorganized and abundant microvessel growth (red arrows)—probably predominantly through a mechanism of intussusceptive angiogenesis—can also be observed (D).
The patient died 15 days after admission due to multiple organ failure.
On autopsy, protein agglomerates with increased expression of surfactant-associated protein A, macrophages, and desquamated alveolocytes were found (Figure 2A) in the corresponding zones with floating structures detected by pCLE (Figure 1B). In areas with partial loss of alveolar structure and filling of the alveoli lumen with moderately fluorescent elements (Figure 1A), histological examination revealed the growth of granulation tissue (Figure 2B).

Protein agglomerates in the lumen of the alveoli with pulmonary macrophages, desquamated type II alveolocytes with the formation of single multinuclear structures (A) (H&E, ×200). Areas of deformed alveolar parenchyma with ingrowth of granulation tissue into the alveolar ducts and alveoli. Features of intraalveolar hemorrhage (B) (H&E, ×100).
Large floating complexes in the alveoli lumen were very similar to pCLE imaging in pulmonary alveolar proteinosis [2]. However, in the context of acute respiratory distress syndrome such structures might be polymorphonuclear neutrophils and macrophages. But the fluorescence intensity of them in our study is visually much higher than presented in the images of Lesur et al. [3], and these cells in our images look like they contain protein-lipid components inside.
In COVID-19 lung tissue, we found a combination of focal alveolar collapse with diffuse hyperplasia of alveolar epithelial type II cells, which express the angiotensin converting enzyme-2 receptor [4]. Apparently, severe acute respiratory syndrome coronavirus 2 primarily damaging pneumocytes, can lead to degradation of the surfactant layer and alveolar atelectasis [5]. In this way, pCLE data may prove the impairment of surfactant production and utilization.
These pathological changes can explain the positive effects of endobronchial surfactant treatment in critically ill covid-19 patients, demonstrated in separate cases [6,7].
Notes
Authors’ Contributions
Conceptualization: Danilevskaya O, Sazonov D, Klypa T. Investigation: Danilevskaya O, Popova E. Writing - original draft preparation: Danilevskaya O, Zabozlaev F. Writing - review and editing: Danilevskaya O, Zabozlaev F. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
No funding to declare.
Acknowledgements
We thank professor Alexander Averyanov, Evgeny Ilyushnikov, Dmitry Panchenkov and Bychinin Mikhail for their help in organizing of the study.
