Barthel’s Index: A Better Predictor for COVID-19 Mortality Than Comorbidities
Article information
Abstract
Background
The most consistently identified mortality determinants for the new coronavirus 2019 (COVID-19) infection are aging, male sex, cardiovascular/respiratory diseases, and cancer. They were determined from heterogeneous cohorts that included patients with different disease severity and previous conditions. The main goal of this study was to determine if activities of daily living (ADL) dependence measured by Barthel’s index could be a predictor for COVID-19 mortality.
Methods
A prospective cohort study was performed with a consecutive sample of 340 COVID-19 patients representing patients from all over the northern region of Portugal from October 2020 to March 2021. Mortality risk factors were determined after controlling for demographics, ADL dependence, admission time, comorbidities, clinical manifestations, and delay-time for diagnosis. Central tendency measures were used to analyze continuous variables and absolute numbers (proportions) for categorical variables. For univariable analysis, we used t test, chi-square test, or Fisher exact test as appropriate (α=0.05). Multivariable analysis was performed using logistic regression. IBM SPSS version 27 statistical software was used for data analysis.
Results
The cohort included 340 patients (55.3% females) with a mean age of 80.6±11.0 years. The mortality rate was 19.7%. Univariate analysis revealed that aging, ADL dependence, pneumonia, and dementia were associated with mortality and that dyslipidemia and obesity were associated with survival. In multivariable analysis, dyslipidemia (odds ratio [OR], 0.35; 95% confidence interval [CI], 0.17–0.71) was independently associated with survival. Age ≥86 years (pooled OR, 2.239; 95% CI, 1.100–4.559), pneumonia (pooled OR, 3.00; 95% CI, 1.362–6.606), and ADL dependence (pooled OR, 6.296; 95% CI, 1.795–22.088) were significantly related to mortality (receiver operating characteristic area under the curve, 82.1%; p<0.001).
Conclusion
ADL dependence, aging, and pneumonia are three main predictors for COVID-19 mortality in an elderly population.
Introduction
Coronavirus disease 2019 (COVID-19) is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a newly emergent coronavirus first recognized in Wuhan, Hubei province, China, in December 2019 [1]. Several systematic reviews and meta-analysis have been published since 2020. They identified several independent risk factors for COVID-19 death, including increasing age [2-11], male sex [5,7-15], hypertension [10,11,13,14,16-18], diabetes mellitus [8,10,11,13-19], obesity [2,6,11], cardiovascular disease (CVD) [10,11,14,16-18,20], cerebrovascular disease [11,14,20], chronic kidney disease [10,11,14,17,20], chronic obstructive pulmonary disease (COPD) [5,8,10-12,16-18,20], cancer [6,8,11,17,18,20-22], dementia [3], frailty [23,24], pneumonia [13], and end-organ damage [13]. Among these risk factors, the most consistently identified predictors were aging, male sex, hypertension, diabetes, CVD, COPD, and cancer. Available evidence still has some limitations: (1) possible ethnic bias; (2) hospital-based studies including patients with different disease severity and patients treated with different levels of care: general wards, respiratory wards, intermediate and intensive care units (ICU); and (3) heterogenous samples (comorbidities and performance status before COVID-19 infection). For these reasons, COVID-19 cases should be split and analyzed considering disease severity, patients’ previous clinical conditions, and the level of care. Some cohort studies have been published accounting for specifically stratified age analysis. However, even in these reports, there were a mix of cases, including ICU and non-ICU patients [25,26]. The main goal of this study was to evaluate the role of activities of daily living (ADL) dependence as a predictor of COVID-19 mortality in a sample of COVID-19 patients treated in a general ward without clinical indications or conditions for ICU admission.
Materials and Methods
1. Study design and sample
This was a prospective cohort study with a consecutive sample. Inpatients referred from tertiary hospitals all over the northern region of Portugal to the Hospital-Escola da Universidade Fernando Pessoa (HE-UFP) (Gondomar, Portugal) from October 2020 to March 2021 were included. All patients had a confirmed COVID-19 diagnosis (positive polymerase-chain reaction from nasal-oropharyngeal swab for SARS-CoV-2). They were treated in a general ward. Patients with clinical indications for intensive care treatment were excluded.
For data analysis, patients were divided into two groups: survivor group (SG) and non-survivor group (NSG). Demographic data, admission time length, comorbidities, evidence of pneumonia (pulmonary consolidation radiologically confirmed by chest X-ray), presence of COVID-19 related symptoms (cough with/without sputum, dyspnea, fever, anosmia, sore throat, myalgia, headache, and/or diarrhea), delay-time from symptoms-to-diagnosis, and in-hospital mortality were analyzed. ADL dependence was measured on the admission using Barthel’s index score. Based on several daily activities (feeding, bathing, grooming, dressing, bowel and bladder control, toilet use capability, transfer from bed to chair and vice-versa, mobility on level-surfaces, and capability to climb stairs), Barthel’s index score has five categories: independent (80–100 points), minimally dependent (60–79 points), partially dependent (40–59 points), very dependent (20–39 points), and totally dependent (<20 points) [27].
Data modeling was performed to identify clinical predictors of mortality in the cohort. It was performed in accordance with the principles of the Declaration of Helsinki.
Informed consent was obtained (written or verbally in case of telephone interview after hospital admission to evaluate mortality up to 3 months after admission). This study was approved by the Institutional Review Board of Universidade Fernando Pessoa Ethics Committee for Health (IRB No. 77, 6/7/2021).
2. Statistical analysis
Continuous variables are described as mean (standard error). Categorical variables are described as absolute numbers and/or proportions. Univariable analysis was performed using Student’s t test for continuous variables and chi-square/Fisher exact test for categorical variables as appropriate. Multivariable analysis was performed using logistic regression (Wald method, p>0.10 for model exclusion/p<0.05 for model inclusion). Age was dichotomized based on a statistical approach to find the best cut-off value to add strength and prediction capability to the model. We decided to analyze ADL dependence based on Barthel’s index original clusters in the univariable analysis and to dichotomize it into two categories (“independent” - including independent and minimally dependent categories; “dependent” - including partially, very, and totally dependent categories) for the multivariable analysis to clearly identify patients with/without ADL dependence. Statistical significance was defined at p<0.05. IBM SPSS version 27 (IBM Corp., Armonk, NY, USA) was used for all statistical analyses.
Results
The total cohort included 340 patients (55.3% females) with a mean age of 80.6±11.0 years. The mortality rate was 19.7% (18.6% in females and 21.1% in males with mean ages of 86.8±8.8 years and 84.8±9.1 years, respectively).
The SG included 273 patients. The NSG included 67 patients. There was no significant difference in sex distribution between the two groups (females: 52.2% in NSG vs. 56.0% in SG, p=0.575).
Deceased patients were significantly older (mean age, 85.8±8.9 years in NSG vs. 79.3±11.1 years in SG; p<0.001).
NSG patients were almost all dependent (98.3% in NSG vs. 77.5% in SG) at certain levels in ADLs according to Barthel’s index score. Partial, very, and total dependencies in ADLs were significantly related to mortality (Table 1). Autonomy or minimal dependency was unrelated to mortality in univariate analysis.
Grouping these latter clusters in the independent group and the former ones in the dependent group revealed that dependency was statistically associated with mortality (odds ratio [OR], 9.603; 95% confidence interval [CI], 2.293–31.544).
Patients were predominantly symptomatic in both groups (86.6% in NSG and 85.8% in SG for any COVID-related symptom, p=0.875). There was no significant difference in the time from symptoms-to-diagnosis between the two groups (1.02±7.5 days in NSG vs. 2.05±4.9 days in SG, p=0.226).
Radiological evidence of pneumonia was more frequent in NSG (78.7% in NSG vs. 60.2% in SG). It significantly increased the risk of mortality (OR, 2.44; 95% CI, 1.26–4.73). Unilateral or bilateral pulmonary affection was not related to an increased risk of death (OR, 0.93; 95% CI, 0.38–2.24).
Comorbidities distribution and their associations with mortality are presented in Table 1. Overall, obesity (body mass index ≥30 kg/m2) (OR, 0.27; 95% CI, 0.10–0.70) and dyslipidemia (OR, 0.36; 95% CI, 0.21–0.64) were associated with survival. Auricular fibrillation (OR, 3.14; 95% CI, 1.71–5.76), end-stage liver disease (liver cirrhosis and failure) (OR, 5.71; 95% CI, 1.25–26.18), and dementia (OR, 2.17; 95% CI, 1.21–3.90) were significantly related to an increased risk for COVID-19 in-hospital death. Survivors had a significant higher mean time length of hospital stay (12.15±7.43 days vs. 8.85±6.65 days, p<0.001). Arterial hypertension, diabetes mellitus, cardiovascular disease, and chronic respiratory diseases were unrelated to COVID-19 mortality in this cohort.
Exploratory analysis helped us define 86-year-old (rounded mean age of the NSG) as the best discriminant cut-off for in-hospital mortality. As such, we decided to categorize age into two groups before modeling: <86 years and ≥86 years. Modeling into a multivariable analysis using logistic regression (Table 2) demonstrated that dyslipidemia (pooled OR, 0.302; 95% CI, 0.147–0.623) was significantly associated with survival.

Multivariable analysis (logistic regression modeling) of in-hospital COVID-19 mortality determinants
Age ≥86 years (pooled OR, 2.239; 95% CI, 1.100–4.559), pneumonia (pooled OR, 3.00; 95% CI, 1.362–6.606), and Barthel’s index score (pooled OR, 6.296; 95% CI, 1.795–22.088) were significantly and independently related to in-hospital COVID-19 mortality.
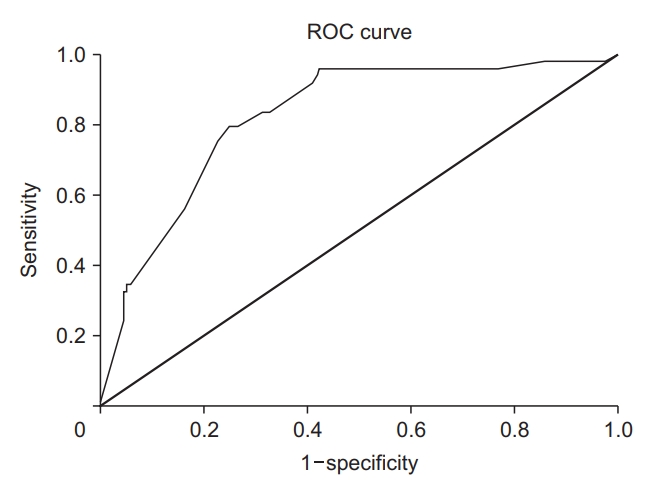
Hosmer and Lemeshow analysis demonstrated that our model had a good fitness (p=0.413) to predict COVID-19 mortality in this cohort of patients. Receiver operating characteristic curve demonstrated an area under the curve of 82.1% (p<0.001) (Figure 1) for death prediction.
Discussion
Our results showed that ADL dependence measured by Barthel’s index score was the most important determinant for COVID-19 mortality in a sample of patients without clinical indications for ICU admission due to age, comorbidities, and/or disease severity.
Aging is a consistent risk factor for COVID-19 death among available trials and revisions [2-11]. This result has several possible explanations such as immune senescence, frailty, and a higher burden of comorbidities. However, this might not be a good explanation since aging is consistently found to be an independent risk factor for death among COVID-19 patients even after controlling for comorbidities. In the first waves of the pandemic, infections and deaths occurred mainly in the elderly population. The present study confirmed such finding. However, we found that ADL dependence was a stronger mortality predictor than individual comorbidities.
Cardiovascular and cerebrovascular diseases are the two main causes of death in the elderly [28] worldwide. Previous data from Kalligeros et al. [29] and Klang et al. [30] have shown an increased risk of COVID-19 mortality in ICU-admitted COVID-19 patients with previous cardiovascular disease and obesity, particularly in younger patients under 50 years old, suggesting that these comorbidities have an important effect on mortality in the most severe and younger cases. However, this might not be the case in the elderly since such risk factors are prevalent in the elderly as seen in our study.
Hypertension, cardiovascular disease (ischemic or valvular), and cerebrovascular disease were unrelated to the risk of death in our cohort. This fact might be explained by the homogeneity of our sample (Table 1): (1) all these comorbidities were highly frequent; (2) young severe patients were not included.
Auricular fibrillation was not independently associated with COVID-19 mortality in our study. This result is different from results of previous studies. Ip et al. [31] reported that auricular fibrillation was associated with COVID-19 mortality, particularly in critically ill patients, consistent with a published meta-analysis [32]. These conflicting results reinforce the need for further specific designed studies for a better understanding of the role of auricular fibrillation in the prognosis of COVID-19 patients.
Frailty has been described as an important risk factor for COVID-19 death, particularly in the elderly [24,33]. Frailty is usually related to old age, ADL dependence, and dementia [34-36]. Interestingly, we found that aging and ADL dependency were independently related to COVID-19 mortality. Dementia lost its significance in the multivariable analysis due to its interaction with ADL dependence (data not shown).
Dyslipidemia was a protective determinant for mortality in our study. The reason for such finding is currently unclear. This might be because dyslipidemia is not associated with malnutrition or an advanced “consumption state” - a frail state. Indeed, malnutrition is usually related to frailty [34-36]. It is not associated with obesity or dyslipidemia.
Obesity was not independently associated with mortality in the current study. Obese patients frequently face difficulties to ventilate. This fact may amplify the effect of obesity in COVID-19 mortality based on other published studies, mainly among cohorts that include ICU patients [37]. Further studies with more homogeneous samples are needed to clarify this issue.
As expected, the presence of pneumonia was related to mortality. Pneumonia is one of the major risk factors for mortality from diseases worldwide [28] in the overall population, including COVID-19 patients [13].
There is conflicting evidence about the role of liver diseases in COVID-19 mortality [13]. We excluded end-stage liver disease from our multivariable model due to terminal stage of such patients represented in the cohort. Liver diseases with milder or moderate presentations should be considered in future studies to clarify their potential role as a deleterious factor in COVID-19 infection.
Respiratory diseases such as asthma, COPD, and interstitial lung diseases were underrepresented in our cohort. The reason for such underrepresentation might be related to an increase in the number of beds in Pulmonology/Oncology wards dedicated to COVID-19 in reference hospitals at the time this study was done. For this reason, conclusions should be taken with caution about these groups of patients.
Our model provided an easy and readily accessible tool to predict mortality in older patients. Age equal to or above 86 years, ADL dependence, and pneumonia were very strong predictors of mortality. There are several models available in the current literature that contain not only clinical variables, but also complex analytical and a priori unmeasurable variables such as admission time [38,39]. Here, we provide a pragmatic tool at the time of diagnosis or admission to predict mortality. It is not designed to predict mortality in all COVID-19 cases. Its application must be considered only in the elderly considering age distribution of our sample.
Many COVID-19 cohort studies have been published with the aim to determine mortality risk factors. They have several sources of heterogeneity such as inclusion of probable cases, mix-analysis of severe and non-severe cases, and inclusion of patients who have died “with” or “from” COVID-19. These facts impose limitations in the generalization of their results. In our study, we only included patients without clinical criteria for intensive care. This inclusion criterion strengthened our findings since possible risk factors and causes of death related to intensive care were excluded (ventilator associated pneumonia, myopathies, insufficient volume-ventilation for obese patients, and so on). Another strength of our study was the homogeneity of the sample. Indeed, comorbidities, age, and sex were evenly distributed. Despite being from a single-center, patients included were representatives from the population all over the northern region of Portugal, which increased the generalizability of our findings.
Our study has some limitations that must be considered. First, its hospital-based nature might have been a natural source of bias. Second, there was an overrepresentation of very old people with several comorbidities. The reason for this was based on the admission criteria to our hospital: confirmed COVID-19 patients without clinical indications or conditions to be admitted to an ICU. There was also an underrepresentation of certain comorbidities such as COPD, asthma, diffuse pulmonary diseases, and cancer. Thus, generalizability of findings for this group of patients must be done with caution.
In our study, ADL dependence was a better predictor of COVID-19 mortality than comorbidities. Together with increasing age and the presence of pneumonia, these three factors were the best predictors for COVID-19 mortality in elderly patients.
Notes
Authors’ Contributions
Conceptualization: da Costa JC, Leite M, Gregório S, Pinto JM. Methodology: da Costa JC, Manso MC, Pinto JM. Formal analysis: da Costa JC, Manso MC, Pinto JM. Data curation: da Costa JC, Manso MC. Software: da Costa JC, Manso MC. Validation: da Costa JC, Manso MC, Gregório S, Leite M, Pinto JM. Investigation: da Costa JC, Manso MC, Leite M, Gregório S, Pinto JM. Writing – original draft preparation: da Costa JC, Manso MC, Leite M. Writing – review and editing: da Costa JC, Manso MC, Gregório S, Leite M, Pinto JM. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
No funding to declare.