Clinical Effectiveness of High-Flow Nasal Cannula in Hypoxaemic Patients during Bronchoscopic Procedures
Article information
Abstract
Background
Bronchoscopy is a useful diagnostic and therapeutic tool. However, the clinical use of high-flow nasal cannula (HFNC) in adults with acute respiratory failure for diagnostic and invasive procedures has not been well evaluated. We present our experiences of well-tolerated diagnostic bronchoscopy as well as cases of improved saturation in hypoxaemic patients after a therapeutic bronchoscopic procedure.
Methods
We retrospectively reviewed data of hypoxaemic patients who had undergone bronchoscopy for diagnostic or therapeutic purposes from October 2015 to February 2017.
Results
Ten patients (44–75 years of age) were enrolled. The clinical purposes of bronchoscopy were for diagnosis in seven patients and for intervention in three patients. For the diagnoses, we performed bronchoalveolar lavage in six patients. One patient underwent endobronchial ultrasonography with transbronchial needle aspiration of a lymph node to investigate tumour involvement. Patients who underwent bronchoscopy for therapeutic interventions had endobronchial mass or blood clot removal with cryotherapy for bleeding control. The mean saturation (SpO2) of pre-bronchoscopy in room air was 84.1%. The lowest and highest mean saturation with HFNC during the procedure was 95% and 99.4, respectively. The mean saturation in room air post-bronchoscopy was 87.4%, which was 3.3% higher than the mean room air SpO2 pre-bronchoscopy. Seven patients with diagnostic bronchoscopy had no hypoxic event. Three patients with interventional bronchoscopy showed improvement in saturation after the procedure. Bronchoscopy was well tolerated in all 10 cases.
Conclusion
This study suggests that the use of HFNC in hypoxaemic patients during diagnostic and therapeutic bronchoscopy procedures has clinical effectiveness.
Introduction
Acute respiratory failure (ARF) has long been a challenge for physicians who perform bronchoscopy for diagnostic or therapeutic purposes. Hypoxaemia is aggravated when implementing bronchoalveolar lavage (BAL) or therapeutic intervention. Many physicians lose their chance to perform bronchoscopy and acquire adequate samples for appropriate treatment. Since undergoing a bronchoscopy can be hazardous to patients with ARF, intubation is preferred and physicians are reluctant to achieve BAL sample. Therefore, bronchoscopic procedures in high-risk patients, especially those with hypoxaemia, have long been a challenge in this field.
Conventional oxygen therapy (low flow O2 system, noninvasive ventilation [NIV]) was known to improve the outcome of hypoxaemic patients. Comparing with high flow nasal cannula (HFNC) group, there were no significant differences in the reintubation rate or length of intensive care unit stay in a meta-analysis study1.
HFNC therapy provides accurate oxygen delivery, wash out of anatomical dead space, and a low-level of positive pressure2. The devices deliver heated and humidified oxygen at a maximum flow rate of up to 60 L/min via nasal cannula, and enable the maintenance of the fraction of oxygen above 95%34.
Miyagi et al.2 presented five cases that underwent BAL with the use of HFNC; the patients tolerated the procedure despite hypoxia and dyspnoea. However, the shortage of cases could not confirm the clinical efficacy and safety of HFNC use during bronchoscopy and BAL. Simon et al.5 reported that NIV was superior to HFNC with regard to oxygenation before, during, and after bronchoscopy in patients with moderate to severe hypoxaemia. However, patients who were stable on HFNC tolerated the bronchoscopy well5.
Few clinical studies have shown the effectiveness of performing bronchoscopy using HFNC in ARF for diagnostic purpose. Moreover, there is no clinical practice that improves oxygenation after performing therapeutic intervention with bronchoscopy. Therefore, we conducted a retrospective study to clarify the clinical effectiveness of high-flow nasal cannula in hypoxaemic patients during a diagnostic and interventional bronchoscopy.
Materials and Methods
This is a retrospective, observational, single center study approved by the ethics committee of the Korea University Guro Hospital. Informed consent was waived due to the retrospective study.
All patients were selected by the Pulmonology Department at a single academic hospital to undergo fiberoptic bronchoscopy and BAL fluid collection for diagnostic purposes or mass removal and cryotheraputic intervention for therapeutic purposes. Patients who had a peripheral arterial pulse oximetry value (SpO2) less than 90% in room air setting were included in this study. Subjects with tracheostomy, requiring home oxygen therapy or mechanical or noninvasive ventilation, nasal or nasopharyngeal disease, not able to clearly express themselves, and pregnant individuals were excluded from the study.
The procedures for all 10 patients were conducted in the same environment. The patients were in the supine position and we administered a mixture of humidified gas warmed by a servo-controlled heated respiratory humidifier (MR730; Fisher & Paykel, Auckland, New Zealand). Baseline room air peripheral saturation (SpO2), heart rate, and respiratory rate were measured before the procedure during spontaneous breathing on room air.
For local anaesthesia, 2% nebulized lidocaine was sprayed twice through the mouth and nostrils. For all patients, a resting period of approximately 5 minutes was needed for the local anaesthesia to fully take effect. Conscious sedation was achieved with midazolam and propofol, which were administered intravenously at maximum doses of 0.1 mg/kg body weight. Fibreoptic bronchoscopy (18-F; Olympus, Corp, Tokyo, Japan) was initiated through a nostril. HFNC was applied through both nostrils. BAL was performed by instilling and aspirating 100 to150 ml of a saline solution (NaCl 0.9%) ample fluid. The highest and lowest saturation (SpO2), heart rate, and respiratory rate were checked during the procedure. The total duration of the bronchoscopy was measured for each patient. At the end of the procedure, the post-bronchoscopy saturation (SpO2) was checked while applying HFNC and on room air after 5 minutes.
Results
1. Patient characteristics
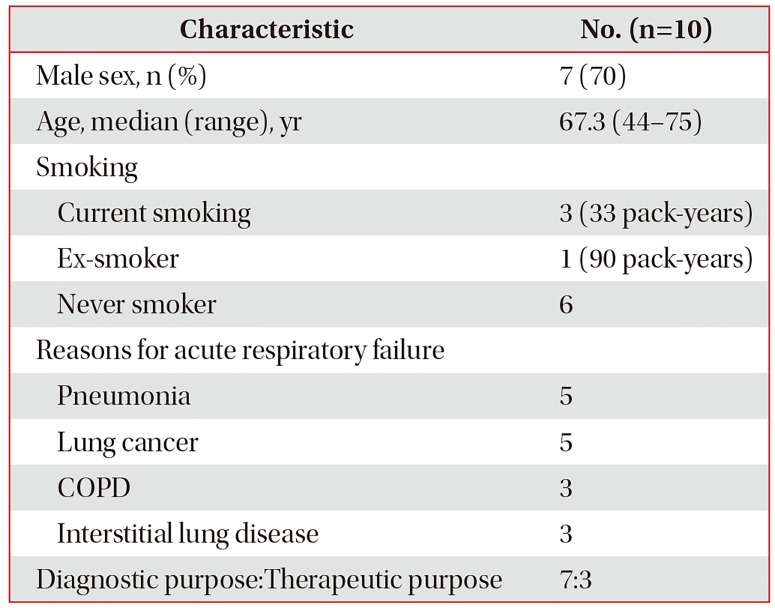
The demographic and clinical characteristics are depicted in Table 1. A total of 10 patients (3 females and 7 males), ranging from 44 to 75 years of age were enrolled. The clinical reasons for bronchoscopy were diagnostic in seven patients and interventional in three patients. Each patient had more than one comorbidity including pneumonia, chronic obstructive pulmonary disease, interstitial lung disease, and lung cancer. Three patients did not have pulmonary function test records, while five patients had a forced expiratory volume in the first second more than 1.0 L and the diffusing capacity of the lungs for carbon monoxide varied between 34% and 60%.
2. Clinical status during bronchoscopic procedures
Seven patients were sedated during bronchoscopy, with close monitoring of heart rate, SpO2, electrocardiogram, and respiratory rate. Baseline room air peripheral saturation (SpO2), heart rate, and respiratory rate were measured before the procedure during spontaneous breathing in room air.
For diagnostic purposes, we performed BAL in five patients with acute pneumonia and one patient with lung cancer. One patient underwent an endobronchial ultrasound transbronchial needle aspiration of a lymph node to evaluate for cancer metastasis. Patients who underwent therapeutic intervention did so for an endobronchial mass or blood clot removal with cryotherapy for bleeding control.
The clinical statuses of the 10 cases that underwent a bronchoscopic procedure with HFNC are shown in Table 2 and vital status before and after bronchoscopy in Table 3. All patients were hypoxic at baseline; therefore, we used HFNC with different settings for each patient. In Figure 1, we compared the SpO2/fraction of inspired oxygen (FiO2) ratio (S/F ratio) prebronchoscopy and postbronchoscopy. Most of the patients undergoing bronchoscopy for diagnosis well tolerated during the procedure by applying HFNC. Three patients undergoing bronchoscopy for therapeutic intervention showed substantial improvement of oxygenation after bronchoscopy on room air after 5 minutes. The mean S/F ratio increase was 35.3.
3. Safety
We were able to discontinue HFNC in all patients after the diagnostic or interventional bronchoscopy were performed. No patient required noninvasive positive pressure ventilation or mechanical ventilation after the completion of the bronchoscopy. HFNC was well tolerated in all 10 cases.
Discussion
Our study was the first to introduce the clinical effectiveness of HFNC for diagnostic and therapeutic interventional bronchoscopy and clarify its usefulness and safety in acute hypoxaemia. HFNC can deliver oxygen at high flow rates (40–60 L/min), permitting a high FiO2. Mucosal injury and patient discomfort is prevented by humidified and heated gas6. Current common clinical applications are to reduce the risk of intubation in patients with moderate or severe hypoxaemia (PaO2:FiO2 <200 mm Hg)7.
The patients in our study can be divided into two groups; one is the diagnostic intervention group and the other the therapeutic intervention group. Among seven patients who had undergone surveillance bronchoalveolar lavage, two patients were found to be influenza A positive by a respiratory virus polymerase chain reaction test. Despite the low yield from bronchoalveolar lavage, the use of HFNC can reduce dyspnoea and discomfort during the procedure, as well as the rate of intubation and duration of hospital stay of the patient during an infection, which can further improve the outcome.
HFNC was applied to both nostrils and the flow in the nostril where the bronchoscope is inserted could have been influenced by the narrowing of the lumen. However, pre and post bronchoscopy SpO2 had no difference.
Lucangelo et al.8 presented a clinical study that determined the effects of HFNC on gas exchange and cardiovascular variables in patients undergoing bronchoscopy and BAL. The included patients had a SpO2 ≥90% and a body mass index ranging from 21 to 3089. In Lucangelo et al.'s study8, 15 patients were applied 60 L/min HFNC during bronchoscopy; however, his study has limitations since it was conducted in patients with mild respiratory dysfunction. Our study has an impact because we showed the efficacy in real clinical scenarios with severe hypoxaemia (SpO2 <90%). All patients maintained SpO2 >91% and no complications were found during and after the procedure. The SpO2 change after the therapeutic intervention, the airway obstruction was solved and resulted in improved room air saturation in three patients (Nos. 7, 9, and 10) (Figure 1).
We carefully suggest an indication for HFNC during bronchoscopy. In an acute infection, where BAL is useful for diagnostic purposes and specimen collection, the use of HFNC during bronchoscopy could be appropriate. An added benefit is the humidified and heated air which can prevent thick secretions and atelectasis. In addition, it is well known that HFNC generates a low level of positive end-expiratory pressure and decreases the dead space of the airway39. However HFNC should not be applied when there is chronic lung disease. Another suggested indication is interventional bronchoscopy in an acutely hypoxaemic state, such as during tumour removal in patients with dyspnoea. Avoiding endotracheal intubation and reducing the need for general anaesthesia in these patients were mentioned as a promising element in case reports and other articles10. All patients were admitted to the general ward rather than the intensive care unit.
There were two limitations to our study. First, the sample size was small. We conducted our investigation at a single center. A multicenter study is required and should be conducted in the near future. Second, since our study was performed retrospectively, setting control group was difficult. Therefore, a prospective case-control study should be conducted in order to determine the clinical indications for diagnostic and therapeutic interventions. In the prospective study, PaO2, pCO2, SpO2, and pH before and after bronchoscopy should all be investigated to clarify the role of HFNC in hypercapnic and hypoxaemic patients.
However, our study has clinical significance in suggesting HFNC indication for hypoxaemic patients before starting the bronchoscopy and to safely finish the procedure. All 10 patients undergone procedures had successful diagnosis and improvement in hypoxaemia. This study can serve as a good background for the prospective case-control study in the near future.
Notes
Authors' Contributions:
Conceptualization: Chung SM, Lee SY.
Formal analysis: Chung SM, Choi JW.
Data curation: Chung SM, Lee SY.
Software: Oh JY, Hur GY.
Administrative support and study supervision: Min KH, Shim JJ, Kang KH.
Writing - original draft preparation: Chung SM, Choi JH.
Writing - review and editing: Chung SM, Lee YS.
Final approval of manuscript: all authors.
Conflicts of Interest: No potential conflict of interest relevant to this article was reported.