Post–Coronavirus Disease 2019 Pulmonary Fibrosis: Wait or Needs Intervention
Article information
Abstract
Coronavirus disease 2019 (COVID-19) has become a major health burden worldwide, with over 450 million confirmed cases and 6 million deaths. Although the acute phase of COVID-19 management has been established, there is still a long way to go to evaluate the long-term clinical course or manage complications due to the relatively short outbreak of the virus. Pulmonary fibrosis is one of the most common respiratory complications associated with COVID-19. Scarring throughout the lungs after viral or bacterial pulmonary infection have been commonly observed, but the prevalence of post– COVID-19 pulmonary fibrosis is rapidly increasing. However, there is limited information available about post–COVID-19 pulmonary fibrosis, and there is also a lack of consensus on what condition should be defined as post–COVID-19 pulmonary fibrosis. During a relatively short follow-up period of approximately 1 year, lesions considered related to pulmonary fibrosis often showed gradual improvement; therefore, it is questionable at what time point fibrosis should be evaluated. In this review, we investigated the epidemiology, risk factors, pathogenesis, and management of post–COVID-19 pulmonary fibrosis.
Introduction
Coronavirus disease 2019 (COVID-19), caused by the novel coronavirus strain severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has continued to spread throughout the world, disrupting our daily lives worldwide since it first broke out in Wuhan, China, in 2019. As of March 2022, more than 450 million cumulative confirmed cases and more than 6 million cumulative deaths have been reported globally due to COVID-19 to date. As the COVID-19 pandemic continues for more than 2 years, the long-term prognosis and clinical course of COVID-19 survivors is attracting attention in addition to the management of the acute phase. Certain people complain of various symptoms for several weeks after acute SARS-CoV-2 infection, which is collectively called post-acute COVID-19 syndrome or “long COVID [1].” Post-acute COVID-19 affects several organ systems; however, the respiratory system is most commonly involved. COVID-19 survivors complain of a spectrum of respiratory symptoms, ranging from mild dyspnea to difficulty weaning from ventilator, due to lung sequelae [2,3].
In COVID-19–related acute respiratory distress syndrome (ARDS) survivors, fatal respiratory complications including failure of withdrawing oxygen supply and home ventilation are frequently observed [4,5]. These complications are presumed to be related to fibrotic lung damage from SARS-CoV-2. Lung sequelae can occur after pulmonary infection; however, fibrotic changes in the lungs after COVID-19 are more frequently observed (range, 20%–70%) [6-14]. However, the clinical course of patients with fibrotic changes induced by COVID-19 may be different from that of patients diagnosed with progressive fibrosing interstitial lung disease (PF-ILD). The radiological features of pulmonary fibrosis generally include distinctive patterns such as architectural distortion, traction bronchiectasis/bronchiolectasis, parenchymal band, reticulation, and honeycombing on computed tomography (CT) [15]. In patients with PF-ILD, fibrotic lesions undergo progression or stabilization, whereas fibrotic lesions gradually improve after pulmonary infection. Therefore, it is challenging to detect pulmonary fibrosis after pulmonary infection. Therefore, previous studies reporting pulmonary fibrosis after COVID-19 favor the term “fibrosis-like pattern” over the term “fibrosis.” In this study, we reviewed post–COVID-19 pulmonary fibrosis with various aspects.
Definition and Epidemiology
The incidence of post–COVID-19 pulmonary fibrosis varies depending on several factors such as population characteristics, fibrosis definition, and observational duration (Table 1). Post–COVID-19 pulmonary fibrosis was mostly characterized by radiological features including reticulation, traction bronchiectasis, parenchymal band, architectural distortion, and honeycombing change, which are also generally considered fibrotic lesions in PF-ILD. A study by Zou et al. [16] revealed that fibrosis, defined as the presence of ground-glass opacity (GGO), evaluated by artificial intelligence-assisted high-resolution computed tomography (HR-CT), was present in 84% of COVID-19 patients at discharge CT. However, the proportion of patients with GGO was 82%, whereas the proportions of patients with reticulation and honeycombing were 30% and 36%, respectively [16]. Since the incidence of post–COVID-19 pulmonary fibrosis differs depending on which lesion is fibrotic, a universal consensus on its definition can help standardize the study.
According to the timing of follow-up, the incidence of post–COVID-19 pulmonary fibrosis was relatively high at midterm (6–9 months) follow-up, whereas it was slightly lower at long-term (more than 1 year) follow-up. Li et al. [13] reported that in 287 patients diagnosed with COVID-19 pneumonia, fibrotic lesion was detected in 88%, 74%, 80%, 68%, and 62% patients, whereas the resolution of fibrotic lesion was observed in 5%, 20%, 18%, 38%, and 49% at 0–30, 31–60, 61–90, 91–120, and >120 days after disease onset. In addition, the proportion of GGO was greatly decreased from 91% in 31–60 days to 65% in >120 days, whereas the proportion of parenchymal bands (95% in 31–60 days and 94% in >120 days) and traction bronchiectasis (14% in 31–60 days and 14% in >120 days) remained stable after the 30-day follow-up [13]. However, in a prospective Italian study (n=118), the proportion of fibrosis-like lesion including reticular pattern with or without honeycombing significantly increased from 55% at baseline to 72%, at the 6-month follow-up [7]. However, in a long-term follow-up Chinese study (n=64), the proportion of reticular abnormalities and traction bronchiectasis did not significantly change from 6 months to 1 year of follow-up [17]. In 209 patients with COVID pneumonia including 22 with ARDS, the most frequently observed predominant CT lesion was GGO during the 1-year follow-up, but the predominant rate of GGO gradually decreased from 83% in discharge CT to 22% after the 1-year follow-up [18]. Meanwhile, the predominance of reticular abnormalities that first appeared at 11% on discharge CT reduced to 3% at 3 months and was maintained during the 1-year follow-up, showing similar rates at the 7-month and 12-month follow-up of 2% and 2%, respectively [18]. Parenchymal bands, another radiological finding of fibrosis, was were confirmed at 0.5%, after the 3-month follow-up; however, it increased to 1% at the 7-month follow-up and remained unchanged until the 1-year follow-up [18]. These findings collectively suggested that as GGOs improve comprehensively, some of them may change to fibrotic lesions because of scarring of the lungs between the 3-month and 6-month follow-up after COVID-19 pneumonia. Once fibrotic lesions occur, they do not improve between the mid- to long-term follow-up, and additional follow-up is needed to identify its accurate incidence. In a 15-year prospective chest CT follow-up of patients with SARS-CoV-1 on April 2003 (n=27), abnormal CT lesions gradually decreased from 2003 to 2018, but remained stable during 2004 and 2018 [19]. This finding suggests that the specific part of pulmonary fibrosis after COVID-19 may be difficult to spontaneously improve without progression.
The characteristics of the included study population is noteworthy. There is a possibility that the incidence of fibrosis is higher in studies that include more patients with severe pneumonia, particularly in patients receiving mechanical ventilation [20]. Therefore, when estimating the incidence of post–COVID-19 pulmonary fibrosis, an approach based on the stratification of fibrosis risk factors, including disease severity, is needed.
Risk Factors
Several risk factors for post–COVID-19 pulmonary fibrosis associated with an individual and disease itself have been proposed in previous studies (Table 2) [6,7,11,12,14,16,20-24]. Aul et al. [20] demonstrated that in COVID-19 patients with available chest CT at 6-week follow-up (n=117), application of invasive ventilation (adjusted odds ratio [OR], 3.48; 95% confidence interval [CI], 1.16–10.49) and persistent breathlessness (adjusted OR, 5.25; 95% CI, 1.86–14.81) were independent risk factors for post–COVID-19 pulmonary fibrosis after adjusting for covariates with a p-value of <0.05 in univariate analysis (male sex, persistent myalgia, peak white blood cell [WBC] count, and high-risk chest X-ray during inpatient COVID-19 admission). Yasin et al. [14] showed that old age (OR, 3.37; 95% CI, 0.76–14.55), CT severity score for opacity estimation (OR, 2.38; 95% CI, 1.18–4.41), presence of consolidation on the initial CT (OR, 1.91; 95% CI, 0.63–4.35), elevated D-dimer level (OR, 1.98; 95% CI, 1.01–10.19), and admission to intensive care unit (ICU) (OR, 6.77; 95% CI, 1.77–25.88) were significantly associated with the occurrence of post–COVID-19 pulmonary fibrosis according to multivariable analysis at median 1.5-month follow-up after discharge in a retrospective Egyptian study (n=21). Zou et al. [16] reported that increased interleukin (IL)-6 level in the acute phase (hazard ratio [HR], 1.081; 95% CI, 1.021–1.144) and decreased serum albumin level (HR, 0.821; 95% CI, 0.734–0.918) were independently associated with post–COVID-19 pulmonary fibrosis in patients with COVID-19 pneumonia (n=248) according to discharge HR-CT. In a prospective 3-month follow-up study (n=173), severe COVID-19 pneumonia (oxygen saturation <90% on room air or signs of severe respiratory distress) according to the World Health Organization classification (OR, 2.40; 95% CI, 1.27–4.51) and the presence of consolidation on the initial CT (OR, 2.84; 95% CI, 1.20–6.73) were significantly associated with the development of post–COVID-19 pulmonary fibrosis according to multivariable analysis [12].
A 6-month follow-up prospective study (n=118) by Caruso et al. [7] revealed that male sex (OR, 0.03; 95% CI, 0.00–0.89), cough (OR, 0.08; 95% CI, 0.01–0.88), lymphocytosis (OR, 0.08; 95% CI, 0.01–0.86), and quantitative chest CT-based well-aerated lung volume (OR, 0.44; 95% CI, 0.01–1.19) were independent protective factors of fibrotic-like changes on multivariable analysis. Yu et al. [22] exhibited that in a retrospective Chines study (n=32; median days from discharge to last follow-up, 9 days), fibrosis was observed in 44% of patients, and patients with fibrosis showed more frequent abnormal radiological features including interstitial thickening, air bronchogram, irregular interface, coarse reticular pattern, and parenchymal bands on worst CT images than those without fibrosis. Although it is not known to be directly related to the development of fibrosis, cigarette smoking and alcohol consumption are potential risk factors for post–COVID-19 pulmonary fibrosis because of their association with the progression of severe pneumonia [25,26]. Overall, post–COVID-19 pulmonary fibrosis can frequently develop in patients with risk factors such as old age, multiple comorbidities, severe pneumonia, and invasive ventilation. Because the previous studies included heterogeneous patients with COVID-19 pneumonia and the definition of fibrosis as well as the observation period was also different, further long-term studies using a common fibrosis criterion are needed to identify risk factors for post–COVID-19 pulmonary fibrosis.
Pathogenesis
The pathogenesis of post–COVID-19 pulmonary fibrosis has not yet been clarified, and multiple pathways are thought to be involved. In idiopathic pulmonary fibrosis (IPF), abnormal wound healing processes, including injury, inflammation, and repair, are key pathways for fibrosis [27]. Acute lung injuries destruct the basement membrane (BM) of the alveolar–capillary barriers and induce inflammatory responses by releasing fibroblast-activating cytokines, chemokines, and growth factors from the recruited inflammatory cells and fibroblasts. Substantial repair processes including angiogenesis and fibroblast activation lead to the deposition of the extracellular matrix (ECM). In normal wound healing response, the repair integrity of the alveolar–capillary barrier BM terminates collagen disposition by activating the fibrolytic process [28]. However, when BM fails to restore its integrity due to persistent or severe injury, fibroblasts and inflammatory pathways are continuously activated, resulting in ECM deposition [29]. These processes collectively contribute to destroyed lung architecture and scarring with fibrosis. Due to an imbalanced wound healing response, excessive deposition of ECM is key to progressive fibrosis in IPF.
1. Inflammatory markers
Severity of pneumonia is regarded as a risk factor for post–COVID-19 pulmonary fibrosis [6,11,12-14]. A study by Huang et al. [11] found that the post–COVID-19 fibrosis group had elevated levels of serum inflammatory markers including WBC, neutrophils, D-dimer, C-reactive protein (CRP), and procalcitonin compared with the non-fibrosis group among patients with COVID pneumonia (n=81). Furthermore, the presence of ARDS, disease severity of pneumonia, or serum inflammatory markers were independent prognostic factors for fibrosis secondary to COVID-19 [6,7,12,14,16]. Therefore, hyperinflammatory status induced by SARS-CoV-2 might be a key pathogenesis of fibrosis. The “cytokine storm” is a phenomenon in which excessive production of proinflammatory cytokines is caused by the hyperimmune reaction of the host to SARS-CoV-2 [30]. The three most crucial mediators of the cytokine storm due to COVID-19 are IL-1, IL-6, and tumor necrosis factor-α (TNF-α). In a Chinese study by Huang et al. [31] (n=41), the initial plasma levels of IL-1β and TNF-α were significantly higher in patients with COVID-19 than in the healthy population, and the level of TNF-α particularly increased in ICU patients (n=13) compared with that in non-ICU patients (n=28). Another Chines study by Chen et al. [32] (n=21) showed that patients with severe COVID-19 had elevated levels of IL-6 and TNF-α as well as increased alanine aminotransferase, lactate dehydrogenase (LDH), CRP, and D-dimer levels and decreased albumin and lymphocyte levels compared with patients with moderate COVID-19. These laboratory findings are similar to the predictive factors for post–COVID-19 pulmonary findings [7,14,16]. TNF-α induces loss of expression on fibroblast Thy-1 surface, leading to myofibroblast differentiation [33]. In addition, TNF-α expression increased in the lungs in IPF [34] and anti–TNF-α antibody diminished pulmonary fibrosis in both bleomycin-induced and silica-induced pulmonary fibrosis in a mouse model [35,36]. In COVID-19, IL-6 level is higher in severe ICU cases and non-survived patients [37-39]. IL-6 stimulates the profibrotic pathway in fibroblasts in patients with IPF, while it enhances apoptosis pathway in normal fibroblasts [40]. Circulating IL-6 levels significantly increased in bleomycin-induced pulmonary fibrosis mice than that in control mice [41]. In vitro, the pathway mediated by soluble IL-6Rα activates proliferation of fibroblasts and production of ECM proteins [42]. Among the cytokines released during COVID-19, profibrotic cytokines were also included, leading to pulmonary fibrosis.
Krebs von den Lungen-6 antigen (KL-6) is one of the biomarkers for ARDS and interstitial lung disease, disrupting the alveolar-capillary barrier [43,44]. Peng et al. [45] reported that the initial serum KL-6 level was significantly higher in patients with fibrosis at discharge CT (n=19) than in patients without fibrosis (n=94). They also observed that serum KL-6 level was elevated in severe cases and not in moderate and mild cases compared with that in controls [45]. Collectively, these findings suggest that patients with severe disease enough to destroy the alveolar-capillary barrier are susceptible to the occurrence of post–COVID-19 pulmonary fibrosis. Therefore, the integrity of the alveolar-capillary barrier may be one of the factors in the pathogenesis of post–COVID-19 pulmonary fibrosis, similar to that in IPF.
2. Renin-angiotensin-aldosterone system
SARS-CoV-2 invades human cells by binding to angiotensin-converting enzyme (ACE) 2, which is a functional receptor mostly expressed on the surface of heart, kidney, and lung cells [46]. Once infected with the virus, the activity of ACE, which is a key regulator of the reninangiotensin-aldosterone system (RAAS), is downregulated by various mechanisms. ACE2 converts angiotensin II to its physiological antagonist angiotensin-(1–7), resulting in vasodilatory, anti-inflammatory, anti-fibrotic, and anti-apoptotic effects [46,47]. Consequently, the activation of vasoconstrictor, proinflammatory, profibrotic, and proliferative effects by the activation of angiotensin II may contribute to post–COVID-19 pulmonary fibrosis. In addition, Xu et al. [48] confirmed that mRNA transcripts of transforming growth factor beta-1 and connective tissue growth factor were significantly elevated in the alveolar epithelium following SARS-CoV-2 binding to ACE2. Because transforming growth factor β (TGF-β) is a potent profibrotic cytokine that induces myofibroblast differentiation, type II alveolar epithelial cell apoptosis, ECM remodeling, it is considered to play a key role in lung fibrogenesis in IPF [49,50]. Therefore, due to SARS-CoV-2 infection, RAAS imbalance may cause post–COVID-19 pulmonary fibrosis by activating the TGF-β pathway.
3. Oxidative stress
Oxidative stress can also contribute to the fibrogenic pathway in post–COVID-19 pulmonary fibrosis. ARDS, which requires high oxygen supply, is a predictive factor for post–COVID-19 pulmonary fibrosis [6]. A case-control study by Farghaly et al. [51] (n=64) demonstrated that patients with COVID-19 undergoing long-term oxygen therapy showed significantly higher CT score, which quantitatively evaluates the extent of abnormal lesion on chest CT than those in patients not undergoing long-term oxygen therapy on chest CT at 1 month (9.6 [oxygen] vs. 6.1 [no-oxygen], p=0.014), and 6 months (4.7 vs. 2.0, p=0.037), after discharge. Hyperoxia induces reactive oxygen species generation in mitochondria [52], with inhibition of oxidative phosphorylation and reduction of adenosine triphosphate level [53], similar to IPF pathogenesis in mitochondria [54]. Meanwhile, in a 4-month follow-up U.S. study (n=76), 10% decrease in an age-adjusted telomere length increased 35% development of fibrotic-like pattern (95% CI, 1.06–1.72) along with sequential organ failure assessment score, LDH level, and duration of invasive ventilation on multivariable analysis [24]. Telomere shortening or mutation is associated with the development of IPF [55]. Taking these points into account, it is possible that post–COVID-19 pulmonary fibrosis may also share a common pathogenetic mechanism with IPF. However, unlike IPF where the abnormal healing process persists due to repeated lung injury, post–COVID-19 pulmonary fibrosis does not progress even if the fibrosis is not significantly improved because additional lung damage does not occur once the active infection subsides. Further clinical studies may be useful to confirm the pathogenesis of post–COVID-19 pulmonary fibrosis.
Clinical Manifestation
Patients with post–COVID-19 pulmonary fibrosis exhibit a wide spectrum of clinical manifestations, ranging from asymptomatic to failure of withdrawing oxygen supply, depending on the extent of fibrosis, follow-up timing, or comorbidities. The most commonly observed symptoms after COVID-19 include fatigue, cough, dyspnea, and sleep disturbance [56,57]. Li et al. [13] reported that at 3-month follow-up, the proportion of symptomatic patients was the highest at 44%, followed by fatigue (21%), cough (15%), and exercise limitation (9%) in patients with fibrosis (n=114). Another 3-month follow-up study revealed that 33.3% patients with had grade ≥2 dyspnea among patients with COVID-19 (n=48) [8]. However, post–COVID-19–related symptoms tend to gradually improve over time [17,51,56,57]. Farghaly et al. [51] demonstrated that the overall mean dyspnea scores for survived patients with post–COVID-19 pulmonary fibrosis at discharge and at the 6-month follow-up were 2.8 and 1.1, respectively. A study by Han et al. [17] also observed that rate of patients with sputum expectoration significantly decreased from 15% in 6 months to 5% in 1 year among patients with severe COVID-19 (n=62). Although statistical significances were not found, the proportion of patients with dry cough (8% in 6 months and 5% in 1 year) and exertional dyspnea (21% in 6 months and 15% in 1 year) also numerically decreased [17].
The most common abnormality in pulmonary function test in post–COVID-19 was impairment of diffusing capacity for carbon monoxide (DLCO) [8,24,58,59]. At 3-month follow-up following discharge, out of the total patients (n=48), 91.3% had fibrosis on chest CT and DLCO was slightly impaired at 61.0% predicted, while other lung functions including forced vital capacity (FVC) and forced expiratory volume per 1 second (FEV1) were within the normal range [8]. At the 4-month follow-up (n=76), patients with fibrotic-like pattern (n=32) also showed decreased DLCO and more weight loss than patients with non-fibrotic-like changes (n=13) or normal CT images (n=31) [24]. Since DLCO was significantly correlated with the extent of traction bronchiectasis (r=−0.49) or reticulation (r=−0.64) on chest CT in patients after COVID-19 [24], DLCO impairment can be frequently observed particularly in patients with post–COVID-19 pulmonary fibrosis. Other lung function abnormalities in FVC, FEV1, and total lung capacity (TLC) can be observed [23,57,60]. In an Italian retrospective study, patients with fibrosis (n=23) had a lower FVC predicted value (50% predicted [fibrosis] vs. 90% predicted [non-fibrosis], p<0.001) compared with patients without fibrosis (n=67) at the 8-week follow-up [60].
Like the symptoms, abnormal lung function also gradually recovered over time [56,57]. The proportion of patients with DLCO of <80% predicted significantly decreased from 31% in 60 days to 21% in 100 days after COVID-19 diagnosis in a prospective, multicenter, observational study (n=145) [56]. Meanwhile, in a Chinese study, the number of patients with impaired lung function including FEV1, FVC, and TLC decreased overtime during 1-year follow-up, particularly that of patients receiving high-flow nasal cannula, invasive or non-invasive mechanical ventilation [57]. However, the proportion of patients with impaired DLCO (<80% predicted) did not change significantly, rather increased numerically from the 6-month to 12-month follow-up [57]. These findings suggested that pulmonary function deterioration due to pulmonary fibrosis may not easily recover during midterm follow-up.
Treatment and Prevention
1. Anti-fibrotics
To date, no effective method has been demonstrated for the treatment or prevention of post–COVID-19 pulmonary fibrosis. Anti-fibrotic agents for the treatment of IPF have been proposed. In IPF, pirfenidone and nintedaninb are recommended to reduce the decline rate of FVC based on clinical trials [61,62]. Nintedanib is a selective tyrosine kinase inhibitor for the receptors of vascular endothelial growth factor, platelet-derived growth factor, and fibroblast growth factor [63], which were significantly elevated in patients with COVID-19 [31]. In a small Japanese interventional study including patients with ventilated COVID-19 pneumonia, the rate of high-attenuation areas on chest CT were not different at the initiation of mechanical ventilation, but significantly lower in patients on nintedanib (n=30) compared with that in controls (n=30), after extubation [64]. In addition, some cases in which fibrosis improved after nintedanib administration have been reported [65,66]. Pirfenidone inhibits profibrotic pathways mostly targeting TGF-β and its downstream pathways, leading to blockage of fibroblast proliferation, myofibroblast transdifferentiation, and collagen deposition [67]. Pirfenidone also has broad anti-inflammatory and anti-oxidative effects by modulating anti-inflammatory cells and cytokines. Zhang et al. [68] revealed that in patients with severe COVID-19 pneumonia, no significant differences in chest CT images between the pirfenidone (n=73) and placebo group (n=73) at 4 weeks of treatment, but pirfenidone group showed lower levels of inflammatory markers including IL-2R, TNF-α, and D-dimer. Other clinical trials showing that anti-fibrotic agents inhibit various profibrotic pathways such as bisamide derivative of dicarboxylic acid, LYT-100, and collagen-polyvinylpyrrolidone and are ongoing [69]. Further long-term clinical trials in different populations can verify the effect of anti-fibrotic agents.
2. Immunomodulatory drugs
There are methods to modify disease-related risk factors to prevent the occurrence of post–COVID-19 pulmonary fibrosis. In particular, because the severity of pneumonia or the occurrence of ARDS is related to fibrosis after COVID-19 [6,7,12,14,16], the administration of corticosteroids or IL-6 receptor inhibitors may be considered to prevent fibrosis by reducing the severity of pneumonia [70,71]. In selective patients with persistent post–COVID-19 interstitial change (n=30), a 6-week prolonged use of corticosteroids improved the clinical course, including lung function and exercise capacity, with tolerability [72]. However, the appropriate dosage and duration of steroids in post–COVID-19 condition remain unclear. In addition, treatment with mesenchymal stem cells, spironolactone, long-term oxygen therapy, and ozone therapy are underway to improve fibrosis.
3. Rehabiltation
Pulmonary rehabilitation can improve lung function, exercise capacity, dyspnea score, and quality of life in patients with ILD [73]. According to a Chinese open randomized controlled study including patients with a post–COVID-19 condition (≥6 months after diagnosis), those who underwent pulmonary rehabilitation (n=36) had a significantly improved lung function, exercise capacity, quality of life, and anxiety/depression assessment score at 6 weeks compared with those before treatment or compared with controls (n=36) [74]. Pulmonary rehabilitation is beneficial in many aspects for patients with a history of ICU admission for respiratory failure, but there is insufficient evidence whether these results can be equally generalized to patients recovering from COVID-19 [75]. Lung transplantation can be an alternative option for end-stage patients with post–COVID-19 pulmonary fibrosis [76,77].
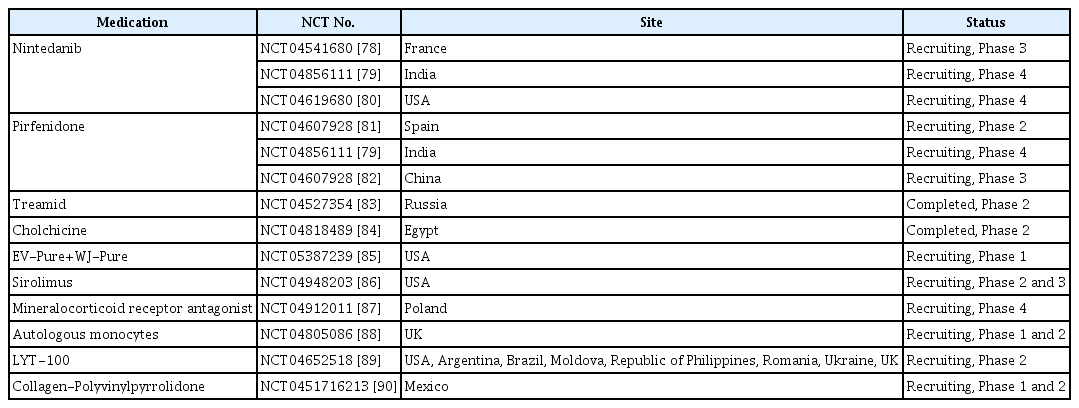
Several ongoing clinical trials have been registered at ClinicalTrials.gov for identifying the efficacy of agents to prevent or diminish secondary fibrogenesis after COVID-19 (Table 3) [78-90]. The efficacy of several trial interventions is being investigated, and the results are expected to be confirmed over a period of months to years.
Conclusion
Approximately 2 years have passed since the outbreak of COVID-19 in 2019, but it is insufficient to evaluate complications. A consensus on the definition of post–COVID-19 pulmonary fibrosis should precede further investigation of its pathogenesis, incidence, and treatment. Furthermore, it is necessary to establish management by observing the results of interventions currently in progress for fibrosis.
Notes
Authors’ Contributions
Conceptualization: Yoon HY, Uh ST. Methodology: Yoon HY. Investigation: Yoon HY. Writing - original draft preparation: Yoon HY. Writing - review and editing: Yoon HY, Uh ST. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
This study was supported by the Soonchunhyang University Research Fund.