Introduction
Surgical resection is the treatment of choice for early stage non-small cell lung cancer (NSCLC). The surgical outcome of these patients is associated with postoperative recurrence. The reported 5-year postoperative recurrence rates of patients with stage I and II NSCLC are approximately 15%-20% and 30%-40%, respectively
1,2. It is impossible to predict which patients are at an increased risk of recurrence after surgical resection based on pathological stage alone. Several studies have shown that the risk factors associated with postoperative recurrence include adenocarcinoma (ADC) histology, the serum carcinoembryonic antigen (CEA) level, tumor differentiation, visceral pleural invasion (VPI), and vessel invasion
1,3,4. In addition, novel predictors of lung cancer, such as the status of epidermal growth factor receptor (EGFR), KRAS mutation and maximal standardized uptake values (SUVs) of tumors on positron emission tomography (PET) were also associated with postoperative outcomes
5,6,7. These results show variety depending on the patient selection or sample size. Another study reported that the risk factors of recurrence were differed between patients with stage I and stage II disease
1.
Currently, the definition of high-risk recurrence is poorly established, and the identification of groups who might benefit from more aggressive adjuvant therapy remains a challenge. An accurate assessment of predictors associated with recurrence is helpful to guide effective adjuvant therapy. Therefore, the aim of this study was to investigate the predictors of recurrence in patients with resected early stage (stage I and II) NSCLC. The patterns of tumor recurrence were also analyzed to identify the risk factors associated with locoregional or distant recurrence.
Discussion
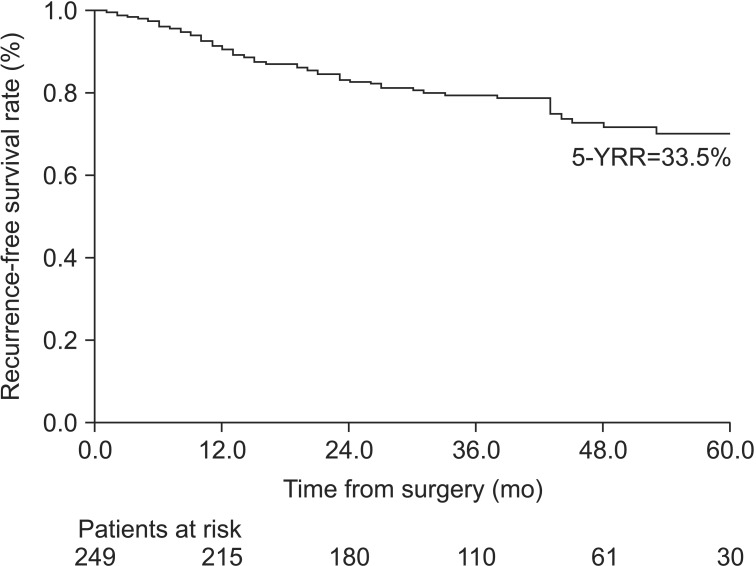
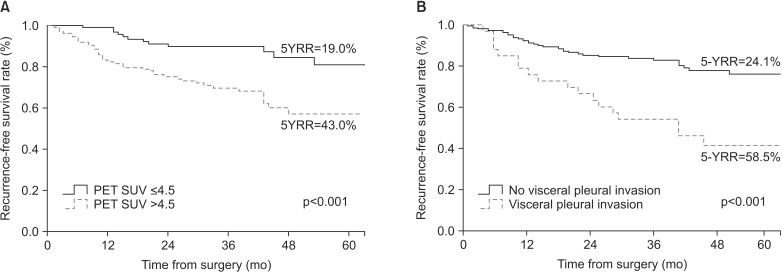
This study investigated the significance of clinical factors other than TNM stage on the time to NSCLC recurrence after complete surgical resection. The presence of VPI and high level of PET SUV of the primary lesion were independent factors associated with postoperative recurrence in early-stage NSCLC patients.
VPI has been mentioned frequently in the various studies concerning the prediction of postoperative recurrence. However, its predictive power has only been confirmed by univariate analysis or appeared to be influenced by the tumor size
3,10. In our study, after adjusting for other confounders, VPI was proven to be an independent predictor of recurrence. VPI is defined as invasion to the surface of the visceral pleura or invasion beyond the elastic layer
11. The degree of VPI was classified into p0-p2 stages, according to the degree of pleural invasion
12. The association of VPI and tumor recurrence has been explained as initial exfoliation in the pleural cavity, followed by preformed stomas that connect the subpleural lymphatics with the pleural space, accounting for the systemic tumor cell dissemination
13,14. As these malignant cells spread through the mediastinal lymphatic vessels, the more frequent incidence of N2 disease is observed in VPI
14. Thus, N2 disease was excluded in our study, and N1 stage was included as a confounding factor in multivariate analysis. The presence of the VPI allows classification of the primary tumor as T2 when it is 3 cm or less in size, according to the seventh edition of the TNM staging system
15. However, recently, some reports have recommended upstaging even if the size of tumor is larger than 3 cm
16,17. It may be inferred from these data that VPI is an important predictor of recurrence and prognosis in earlystage NSCLC, independent of the tumor size. Therefore, adjuvant therapy should be more likely to be recommended in patients with VPI.
Although PET SUV has not been clearly identified in association with the recurrence of cancer, several studies have reported that SUV is a prognostic factor for recurrence
7,18,19. This may be because the more metabolically active is the tumor, the worse the outcome will be. However, each of the studies reported different SUV cut-off values
20,21. The SUV values were determined by plasma glucose levels, tumor size, and type of neoplasm
7,22. In particular, regarding the relationship between SUV and the tumor histopathology, a previous study showed that the SUV was significantly higher in SCC than ADC
19,23. In our study, the cut-off SUV value of 4.5 was determined by the ROC of all patients, and the same cut-off value was applied to both ADC and SCC. We performed subgroup analysis, which applied different cut-off values of SUV according to the histologic subtypes. The median value of SUV was significantly different between these groups (p<0.001): ADC, 3.6 (IQR, 2.1-6.5); SCC, 9.9 (IQR, 6.5-12.5). In the ADC group (n=158), a PET SUV ≥2 was significantly associated with 5-year recurrence by the Kaplan-Meier with log-rank test. No one has shown recurrence in patients with SUV <2. In addition, the proportion of GGO in the total patients was 20.9%. The result was not changed after excluding the patients with GGO. However, in the SCC group, there was no significant association between a high level of PET SUV and tumor recurrence with a cut-off value of 9 or 10. This suggests that PET SUV may play an important role in the outcomes of ADC patients, but not in those of SCC patients. However, studies concerning the relationship between PET SUV and the recurrence of earlystage NSCLC, particularly ADC patients, are rare. Thus, further study is needed to confirm our findings.
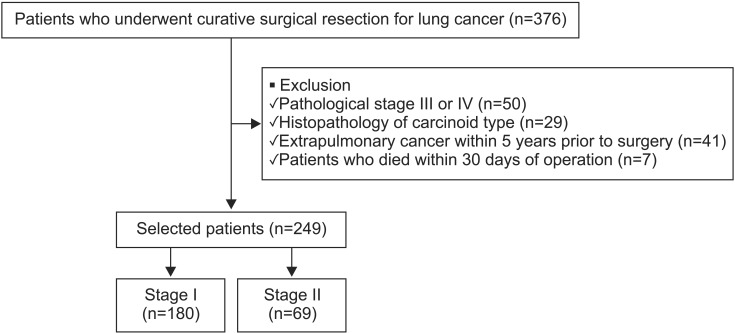
In the analysis of stage I patients, VPI and positive margin status were independent risk factors of recurrence. However, in stage II patients, only ADC was associated with recurrence by univariate analysis. These results were similar to a previous study that found the risk factors differed between patients with stages I and II disease
1. In patients with AIS or MIA, disease-specific survival was almost 100% after complete resection. However, invasive ADC, particularly micropapillary or solid-predominant, has a more aggressive behavior that favors early distant metastasis
24. In our study, the proportion of GGO was significantly high in stage I (38.1%) compared with that in stage II patients (6.3%) (p<0.001). Additionally, AIS or MIA were included among stage I, but not among stage II patients. Among patients with invasive ADC, the proportion of micropapillary or solid-predominant subtypes was higher in stage II (28.0%) than in stage I patients (12.8%) (with no statistical significance). These results indicate that more aggressive subtypes would be included in stage II than in stage I disease. Therefore, ADC could be a poor prognostic factor in stage II, but not stage I, disease, instead of other factors, such as VPI, which played a more important role in recurrence for stage I patients.
We also analyzed the patterns of recurrence and factors associated with locoregional or distant recurrence. Although there is variation in the literature, it is generally believed that the risk of local recurrence for patients with early-stage NSCLC is low
9,25. Several studies have explained the finding as follows. First, local recurrence may be missed because it is difficult to detect from an isolated lymph node or stump site recurrence
9. In addition, concurrent local and distant recurrences are common, and they are regarded as distant recurrences. However, it is possible that local recurrence occurred before and may not be detected until distant recurrence developed. Another explanation is that the definition of local failure varies widely. Some studies defined local failure as occurring only in the ipsilateral lung and mediastinum
9. However, the contralateral lymph nodes were classified as local failure in other studies
26. Our study showed that the frequency of distant recurrence appeared to be similar to that of local recurrence. This is because the most common recurrence site was the lymph nodes and contralateral mediastinal recurrence was regarded as local failure.
Previous studies reported that clinical factors associated with local or distant recurrence were different from each other
3,26. Factors associated with local recurrence included diabetes and LVI. On the other hand, distant recurrence was associated with histology type, such as ADC, performance of pneumonectomy, and advanced stage in other studies
26. However, evidence to confirm these findings is lacking. In our study, only VPI was shown to be associated with both local and distant recurrence. Involvement of the N1 lymph nodes was an independent risk factor of locoregional recurrence. Because the number of patients with recurrence was small and the duration of the follow-up was short, the interpretation of these results should be limited. Information from larger population studies is warranted to determine the factors associated with the patterns of recurrence in early-stage NSCLC.
There were several limitations to this study. First, its retrospective nature resulted in various biases, and study was performed at a single center. Thus, the patient characteristics may not be representative. Second, because the follow-up duration was insufficient, we could not perform an analysis of the overall survival rate of patients. Third, the rate of a positive resection margin was higher (5.2%) compared with that in previous studies
3,9, and postoperative radiotherapy or re-surgery was only performed to two patients.
Another limitation of our study is that recurrence group had a higher rate of receiving adjuvant chemotherapy compared with no-recurrence group (
Table 1), and adjuvant chemotherapy was associated with 5-year recurrence in multivariate analysis (with no statistical significance) (
Table 2). When we analyzed the clinical characteristics of patients receiving adjuvant chemotherapy compared with non-receiving patients in stage I disease, they had a higher rate of a positive margin, mod-poorly differentiated carcinoma, and
KRAS mutation. These factors are more likely to influence tumor recurrence, and suggest that patients with a high risk of recurrence are likely to have received adjuvant chemotherapy in stage I disease. In addition, adjuvant chemotherapy is standard therapy in stage II disease; however, only 47.8% of stage II patients in our study received adjuvant chemotherapy. Therefore, this must be considered in the interpretation of the results. Despite these limitations, our study presented current issues regarding predictors of recurrence after curative resection and which patients will benefit from adjuvant therapy.
In summary, our study shows that the presence of the VPI and a preoperative high level of PET SUV were independent factors associated with postoperative recurrence in earlystage NSCLC patients. Although the risk factors associated with recurrence would differ between stage I and II, patients with these risk factors should be considered for more aggressive adjuvant therapy. Prospective, large-scale multicenter studies with longer follow-up periods are required to identify the predictors of postoperative outcomes.





 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation



